Which Element Below Is Least Reactive
listenit
Apr 02, 2025 · 5 min read
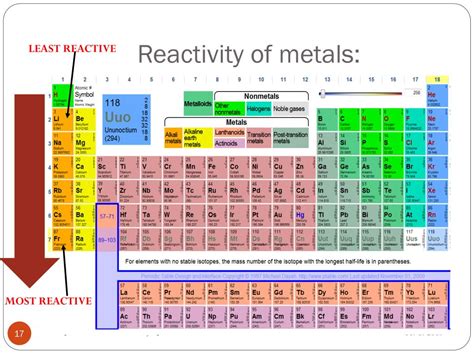
Table of Contents
Which Element Below is Least Reactive? Understanding Reactivity in the Periodic Table
The question, "Which element below is least reactive?" hinges on understanding chemical reactivity. It's not a simple yes or no answer; it requires a deep dive into the periodic table and the factors influencing an element's desire (or lack thereof) to participate in chemical reactions. Reactivity isn't a single, universally applicable value; it depends on the specific conditions—the presence of other elements, temperature, pressure, and more. However, we can establish a general hierarchy based on well-established chemical principles.
Understanding Chemical Reactivity
Chemical reactivity refers to an element's tendency to undergo chemical changes, specifically to form chemical bonds with other elements. This tendency stems from the arrangement of electrons in an atom's outermost shell, known as the valence shell. Atoms strive for stability, often achieved by having a full valence shell—a configuration similar to noble gases. Elements readily react to achieve this stable electron configuration.
Factors Affecting Reactivity
Several factors influence an element's reactivity:
-
Electron Configuration: The number of valence electrons directly impacts reactivity. Elements with nearly full or nearly empty valence shells tend to be more reactive than those with half-full or completely full shells.
-
Electronegativity: Electronegativity measures an atom's ability to attract electrons in a chemical bond. Highly electronegative elements readily attract electrons, often forming strong bonds and exhibiting high reactivity.
-
Ionization Energy: This is the energy required to remove an electron from an atom. Elements with low ionization energies readily lose electrons, increasing their reactivity.
-
Atomic Radius: The size of an atom influences reactivity. Larger atoms have valence electrons further from the nucleus, making them easier to remove or share, thus increasing reactivity.
-
Metallic Character: Metals tend to lose electrons readily, displaying high reactivity. Non-metals, conversely, often gain electrons, also leading to significant reactivity (though in different ways).
The Least Reactive Elements: The Noble Gases
The least reactive elements are the noble gases, located in Group 18 of the periodic table. These include helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), and radon (Rn). Their exceptional stability arises from their complete valence electron shells. They have no strong tendency to gain, lose, or share electrons to achieve a more stable configuration because they already possess it.
Why are Noble Gases Inert?
The noble gases' full valence shells make them incredibly unreactive. They are often referred to as inert gases due to their minimal participation in chemical reactions under normal conditions. The energy required to force them to react is exceptionally high, making spontaneous reactions extremely rare.
However, it's crucial to note that even noble gases can participate in chemical reactions under extreme conditions, such as extremely low temperatures or high pressures, or when reacting with highly reactive species. For example, xenon, being the largest noble gas atom, has been shown to form compounds under specific laboratory conditions. But these are exceptions to the rule. Under typical circumstances, they remain remarkably unreactive.
Comparing Reactivity Across the Periodic Table
To fully appreciate the low reactivity of noble gases, it's helpful to compare them with other elements:
Alkali Metals (Group 1)
Alkali metals, like lithium (Li), sodium (Na), and potassium (K), are highly reactive. They have only one valence electron, readily losing it to form a +1 ion and achieve a stable electron configuration. Their reactivity increases as you move down the group due to increasing atomic size and decreasing ionization energy.
Alkaline Earth Metals (Group 2)
Alkaline earth metals, like beryllium (Be), magnesium (Mg), and calcium (Ca), are also reactive, though less so than alkali metals. They have two valence electrons, readily losing them to form +2 ions.
Halogens (Group 17)
Halogens, such as fluorine (F), chlorine (Cl), and bromine (Br), are highly reactive non-metals. They have seven valence electrons and readily gain one electron to form a -1 ion, achieving a stable octet. Their reactivity decreases as you move down the group.
Transition Metals
Transition metals exhibit variable reactivity depending on their electronic configuration and oxidation states. Some are quite reactive, while others are relatively inert.
Identifying the Least Reactive Element in a Given Set
When presented with a list of elements, the key to identifying the least reactive one is to look for the element closest to the noble gases in terms of electron configuration. Elements with full or nearly full valence shells will exhibit the lowest reactivity.
Example:
Let's say you are given the following elements: sodium (Na), chlorine (Cl), argon (Ar), and oxygen (O). The least reactive element would be argon (Ar) because it's a noble gas with a completely filled valence shell. Sodium and chlorine are highly reactive, while oxygen is moderately reactive.
Practical Applications of Unreactive Elements
The low reactivity of noble gases makes them valuable in various applications:
-
Helium: Used in balloons, airships, and cryogenics due to its low density and inertness.
-
Neon: Used in neon signs due to its characteristic red glow when an electric current is passed through it.
-
Argon: Used as an inert atmosphere in welding and other industrial processes to prevent oxidation.
-
Krypton and Xenon: Used in specialized lighting applications due to their unique spectral properties.
Conclusion: Reactivity – A Spectrum, Not a Binary
While noble gases are generally considered the least reactive elements, reactivity is a spectrum, not a simple binary on/off switch. Even noble gases can participate in reactions under specific conditions. Understanding the factors influencing reactivity – electron configuration, electronegativity, ionization energy, and atomic radius – is crucial for predicting and understanding chemical behavior. By focusing on the electron configuration and the stability conferred by a full valence shell, you can reliably identify the least reactive element in any given set.
Latest Posts
Latest Posts
-
A Square Is Always A Parallelogram
Apr 03, 2025
-
What Percent Of 8 Is 7
Apr 03, 2025
-
One Degree Celsius Is Equal To How Many Degrees Fahrenheit
Apr 03, 2025
-
What Is The Diploid Number Of Chromosomes In Corn
Apr 03, 2025
-
What Is It Called When Liquid Changes To Gas
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Which Element Below Is Least Reactive . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
