What Is It Called When Liquid Changes To Gas
listenit
Apr 03, 2025 · 6 min read
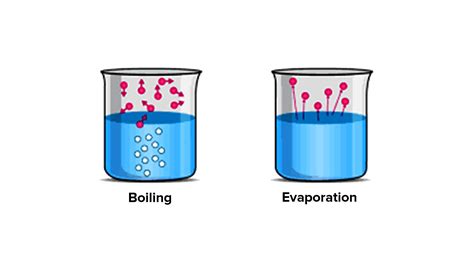
Table of Contents
What is it called when liquid changes to gas? Understanding Vaporization and its Types
The transformation of a liquid into a gas is a fundamental process in nature and science, crucial in everything from weather patterns to industrial processes. This phenomenon, broadly termed vaporization, encompasses several specific types of changes, each with its own characteristics and underlying mechanisms. Understanding these differences is key to grasping the complexities of phase transitions and their implications.
Understanding Vaporization: From Liquid to Gas
Vaporization, at its core, is the process where a liquid transitions to a gaseous state. This happens when sufficient energy, typically in the form of heat, is supplied to overcome the intermolecular forces holding the liquid molecules together. These forces, depending on the substance, vary in strength. Stronger intermolecular forces require more energy to break, resulting in higher boiling points.
The process of vaporization isn't uniform; it manifests in several distinct ways, each with its own set of conditions and characteristics. Let's delve into the different types of vaporization:
1. Evaporation: A Gradual Transition
Evaporation is the most commonly observed type of vaporization. It's a process where liquid changes into gas at temperatures below the liquid's boiling point. It occurs at the surface of a liquid, as molecules with sufficient kinetic energy escape into the surrounding atmosphere. Think of a puddle drying up in the sun, or a spilled drink slowly disappearing.
Factors Influencing Evaporation:
- Temperature: Higher temperatures provide molecules with more kinetic energy, leading to faster evaporation.
- Surface area: A larger surface area exposes more liquid molecules to the atmosphere, increasing the rate of evaporation.
- Humidity: High humidity means the air is already saturated with water vapor, reducing the rate of evaporation. The air's capacity to hold more water vapor is reduced.
- Air movement: Wind or air currents whisk away evaporated molecules, preventing them from returning to the liquid and speeding up the process.
- Nature of the liquid: Different liquids have different vapor pressures, influencing how readily they evaporate. Substances with high vapor pressure evaporate more quickly.
2. Boiling: A Violent Transformation
Boiling, also known as ebullition, is a more dramatic form of vaporization. It occurs when a liquid's temperature reaches its boiling point, the temperature at which the vapor pressure of the liquid equals the surrounding atmospheric pressure. At this point, vapor bubbles form throughout the liquid's bulk, not just at the surface, and rise rapidly to the surface, leading to vigorous bubbling and a rapid transition to the gaseous phase.
Understanding Boiling Point:
The boiling point isn't a fixed value; it's dependent on the external pressure. At higher altitudes, where atmospheric pressure is lower, the boiling point of a liquid decreases. This is why water boils at a lower temperature on mountaintops. Conversely, at higher pressures, the boiling point increases. Pressure cookers utilize this principle to cook food faster at higher temperatures.
3. Sublimation: Bypassing the Liquid Phase
While not strictly a form of vaporization in the traditional sense, sublimation is a noteworthy phase transition. It's the process where a solid directly transforms into a gas without passing through the liquid phase. Dry ice (solid carbon dioxide) is a classic example. At atmospheric pressure, it transforms directly into carbon dioxide gas, creating a visually striking effect of "smoke."
Factors Affecting Sublimation:
Similar to evaporation, factors influencing sublimation include temperature and pressure. Higher temperatures and lower pressures favor sublimation. The nature of the solid also plays a role; some solids sublime more readily than others.
The Role of Heat in Vaporization: Understanding Latent Heat
Heat plays a pivotal role in vaporization, supplying the energy needed to overcome the intermolecular forces holding the liquid together. The heat absorbed during vaporization is termed latent heat of vaporization. This heat doesn't raise the temperature of the liquid; instead, it's used to break the bonds between molecules, allowing them to transition into the gaseous phase.
Latent Heat and its Significance:
The latent heat of vaporization is a specific property of each substance, indicating the amount of energy required to vaporize a unit mass of that substance at its boiling point. This property is crucial in various applications, including cooling systems and industrial processes. For example, sweating utilizes the latent heat of vaporization of water to cool the body.
Practical Applications of Vaporization
Vaporization is a fundamental process with numerous applications across various fields:
1. Industrial Processes: Distillation and Refining
Distillation, a technique based on differences in boiling points, is widely used in the chemical industry to separate components of mixtures. Crude oil refining, for instance, relies heavily on distillation to separate different fractions, each with distinct boiling ranges.
2. Cooling Systems: Refrigeration and Air Conditioning
Refrigeration and air conditioning systems utilize vaporization and condensation cycles to transfer heat. Refrigerants, liquids with low boiling points, vaporize at low temperatures, absorbing heat from the surroundings, and then condense at higher temperatures, releasing the heat elsewhere.
3. Weather Patterns: Evaporation, Condensation, and Precipitation
Evaporation plays a crucial role in the water cycle. Water evaporates from oceans, lakes, and rivers, forming clouds through condensation, ultimately leading to precipitation. Understanding vaporization is thus vital in comprehending weather patterns and climate.
4. Power Generation: Steam Turbines
Steam turbines utilize the vaporization of water to generate electricity. High-pressure steam, produced by boiling water, drives turbines, converting heat energy into mechanical energy, which is then transformed into electricity.
Vapor Pressure: A Key Concept
Vapor pressure is the pressure exerted by a vapor in equilibrium with its liquid phase at a given temperature. It represents the tendency of a liquid to evaporate. A higher vapor pressure indicates a greater tendency to evaporate. Vapor pressure increases with temperature, as more molecules gain enough energy to escape the liquid phase.
Factors Affecting Rate of Vaporization: A Deeper Dive
Several factors intricately influence the rate of vaporization beyond those already mentioned:
- Molecular weight: Heavier molecules generally have lower vapor pressures and evaporate more slowly.
- Intermolecular forces: Stronger intermolecular forces (hydrogen bonding, dipole-dipole interactions, London dispersion forces) lead to lower vapor pressures and slower evaporation.
- Concentration: The concentration of a substance in a solution can affect its evaporation rate. A higher concentration can sometimes lead to slower evaporation, depending on the interactions between the solute and solvent.
- Surface impurities: Impurities on the liquid's surface can hinder evaporation by altering the surface tension.
Conclusion: Vaporization – A Ubiquitous Process
Vaporization, in its various forms, is a ubiquitous process fundamental to many natural and industrial phenomena. Understanding the different types of vaporization, the influence of various factors, and the related concepts like latent heat and vapor pressure is crucial in numerous fields, ranging from meteorology and chemistry to engineering and environmental science. The ongoing research in this area continues to refine our understanding of this essential phase transition and its implications. By appreciating the complexities and nuances of vaporization, we gain a deeper insight into the world around us and the processes that shape our environment.
Latest Posts
Latest Posts
-
100 Yards On A Football Field
Apr 04, 2025
-
A Consumer That Eats Only Plants
Apr 04, 2025
-
The Amount Of Energy Required To Raise The Temperature
Apr 04, 2025
-
38 Out Of 40 Is What Percent
Apr 04, 2025
-
Which Polynomial Represents The Sum Below
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about What Is It Called When Liquid Changes To Gas . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
