The Amount Of Energy Required To Raise The Temperature
listenit
Apr 04, 2025 · 6 min read
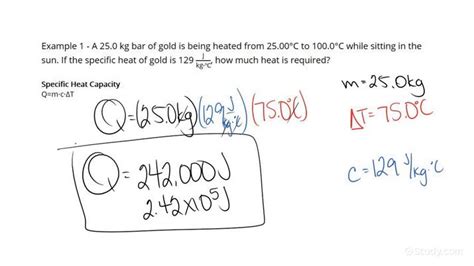
Table of Contents
The Amount of Energy Required to Raise the Temperature: A Deep Dive into Specific Heat Capacity
Understanding the amount of energy needed to change an object's temperature is fundamental to numerous scientific fields, from thermodynamics and engineering to meteorology and cooking. This crucial concept hinges on specific heat capacity, a property that dictates how readily a substance absorbs or releases heat. This article delves into the intricacies of specific heat capacity, exploring its definition, influencing factors, applications, and the calculations involved in determining the energy required for temperature changes.
Defining Specific Heat Capacity
Specific heat capacity, often denoted as 'c', quantifies the amount of heat energy required to raise the temperature of one unit of mass of a substance by one degree Celsius (or one Kelvin). It's expressed in units of Joules per kilogram per Kelvin (J/kg·K) or Joules per gram per degree Celsius (J/g·°C). The key takeaway is that different substances require varying amounts of energy to achieve the same temperature change. Water, for instance, boasts a remarkably high specific heat capacity, meaning it absorbs a significant amount of heat for a relatively small temperature increase. This property is crucial for regulating Earth's climate and maintaining stable temperatures in aquatic environments.
Why Specific Heat Capacity Varies
Several factors influence a substance's specific heat capacity:
-
Molecular Structure and Bonding: The arrangement of atoms and molecules within a substance directly impacts how efficiently it can absorb and distribute thermal energy. Substances with complex molecular structures, involving numerous bonds and interactions, often exhibit higher specific heat capacities. This is because the energy is distributed across many vibrational modes.
-
Intermolecular Forces: Stronger intermolecular forces (like hydrogen bonds in water) require more energy to overcome, leading to higher specific heat capacities. These forces restrict the movement of molecules, requiring more energy to increase their kinetic energy and thus raise the temperature.
-
Phase of Matter: The phase of a substance (solid, liquid, or gas) dramatically affects its specific heat capacity. Generally, solids have lower specific heat capacities than liquids, which in turn have lower specific heat capacities than gases. This difference stems from the varying degrees of freedom available to the molecules in each phase. Gaseous molecules have more freedom of movement, necessitating more energy to increase their kinetic energy.
-
Temperature: While often considered constant, specific heat capacity can actually vary slightly with temperature. This variation is usually minor, especially within a relatively narrow temperature range, but becomes more significant at extreme temperatures or phase transitions.
The Formula: Calculating Energy Change
The relationship between energy change (ΔQ), mass (m), specific heat capacity (c), and temperature change (ΔT) is expressed by the following equation:
ΔQ = mcΔT
Where:
- ΔQ represents the heat energy transferred (in Joules). A positive ΔQ indicates heat absorbed, while a negative ΔQ signifies heat released.
- m is the mass of the substance (in kilograms or grams).
- c is the specific heat capacity of the substance (in J/kg·K or J/g·°C).
- ΔT is the change in temperature (in Kelvin or degrees Celsius). ΔT = T<sub>final</sub> - T<sub>initial</sub>
This formula provides a straightforward way to calculate the energy required to change a substance's temperature. For example, to determine the energy needed to heat 1 kg of water by 10°C (assuming a constant specific heat capacity of 4186 J/kg·°C), the calculation would be:
ΔQ = (1 kg)(4186 J/kg·°C)(10°C) = 41860 J
This indicates that 41860 Joules of energy are required to heat 1 kg of water by 10°C.
Applications of Specific Heat Capacity
The concept of specific heat capacity has far-reaching applications in numerous fields:
1. Climate Regulation:
Water's high specific heat capacity plays a vital role in moderating Earth's climate. Large bodies of water absorb vast amounts of solar energy without experiencing drastic temperature fluctuations. This prevents extreme temperature swings and creates more stable environments. Coastal regions, for example, tend to have milder climates than inland areas due to the influence of the ocean's high specific heat capacity.
2. Engineering and Material Science:
Engineers utilize specific heat capacity data to design and optimize systems involving heat transfer. Choosing materials with appropriate specific heat capacities is crucial for applications like heat sinks in electronics, heat exchangers in power plants, and thermal insulation in buildings. The ability to predict and control temperature changes is vital in many engineering applications.
3. Meteorology and Atmospheric Science:
Understanding the specific heat capacities of different atmospheric gases (like nitrogen, oxygen, and carbon dioxide) helps meteorologists and climate scientists model atmospheric processes, predict weather patterns, and analyze the effects of climate change. These gases' heat capacities influence the distribution of heat energy in the atmosphere, impacting temperature gradients and weather systems.
4. Cooking and Food Science:
In cooking, specific heat capacity influences how quickly foods heat up or cool down. Understanding the specific heat capacities of different ingredients allows chefs to control cooking times and temperatures accurately, ensuring optimal food texture and flavor. The rapid heating of some ingredients compared to others directly relates to their varying specific heat capacities.
5. Medical Applications:
Specific heat capacity is relevant in medical applications such as hyperthermia cancer treatment. This treatment method involves selectively heating cancerous tissues to destroy them. Accurate calculation of energy requirements based on the specific heat capacity of the tissue is vital for effective and safe treatment.
Advanced Considerations and Limitations
While the formula ΔQ = mcΔT provides a useful approximation, several factors need to be considered for more accurate calculations:
-
Phase Transitions: The formula doesn't account for phase transitions (melting, boiling, etc.). During phase transitions, energy is absorbed or released without a change in temperature. Latent heat, a separate concept, describes the energy involved in these phase changes.
-
Temperature Dependence: As mentioned, specific heat capacity can vary with temperature. For more precise calculations at extreme temperatures, it's necessary to account for this variation.
-
Pressure Dependence: Specific heat capacity can also be slightly influenced by pressure. High-pressure environments can affect the interactions between molecules, leading to changes in heat capacity.
-
Non-uniform Temperature Distribution: The formula assumes a uniform temperature distribution throughout the substance. However, in real-world scenarios, temperature gradients often exist, requiring more complex methods for accurate heat transfer calculations.
Conclusion: A Crucial Thermodynamic Property
Specific heat capacity is a fundamental concept in thermodynamics with far-reaching applications across diverse scientific and engineering disciplines. Its influence extends from global climate patterns to the design of everyday objects and medical procedures. Understanding how specific heat capacity affects energy transfer is essential for accurate prediction and control of temperature changes in various systems. While the basic formula provides a helpful starting point, a comprehensive understanding necessitates considering the limitations and applying more sophisticated methods in complex situations. Further exploration of latent heat and the temperature and pressure dependence of specific heat capacity will provide an even deeper insight into this crucial thermodynamic property.
Latest Posts
Latest Posts
-
How Many Unpaired Electrons Does Manganese Have
Apr 04, 2025
-
Water Is Made Up Of Which Two Elements
Apr 04, 2025
-
What Is 9 Percent Of 50
Apr 04, 2025
-
Distance From Earth To Pluto In Light Years
Apr 04, 2025
-
Molar Mass Of Copper 2 Sulfate
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about The Amount Of Energy Required To Raise The Temperature . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
