Molar Mass Of Copper 2 Sulfate
listenit
Apr 04, 2025 · 5 min read
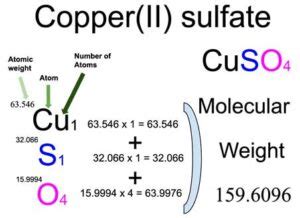
Table of Contents
Understanding the Molar Mass of Copper(II) Sulfate: A Comprehensive Guide
Copper(II) sulfate, a vibrant blue crystalline compound with the chemical formula CuSO₄, finds extensive applications in various fields, from agriculture and industry to medicine and laboratory settings. Understanding its molar mass is crucial for accurate stoichiometric calculations, solution preparation, and various chemical analyses. This comprehensive guide delves deep into the concept of molar mass, specifically focusing on copper(II) sulfate, exploring its calculation, significance, and practical applications.
What is Molar Mass?
Molar mass, often referred to as molecular weight, represents the mass of one mole of a substance. A mole is a fundamental unit in chemistry, defined as the amount of a substance that contains Avogadro's number (approximately 6.022 x 10²³) of constituent particles, whether atoms, molecules, or ions. The molar mass is expressed in grams per mole (g/mol). It's a crucial conversion factor bridging the microscopic world of atoms and molecules to the macroscopic world of grams and kilograms that we measure in the lab.
Calculating Molar Mass: A Step-by-Step Approach
The calculation of molar mass involves summing the atomic masses of all the atoms present in a molecule or formula unit. These atomic masses are typically found on the periodic table of elements. For copper(II) sulfate (CuSO₄), the calculation is as follows:
- Copper (Cu): One copper atom has an atomic mass of approximately 63.55 g/mol.
- Sulfur (S): One sulfur atom has an atomic mass of approximately 32.07 g/mol.
- Oxygen (O): Four oxygen atoms, each with an atomic mass of approximately 16.00 g/mol, contribute a total of 4 * 16.00 = 64.00 g/mol.
Therefore, the molar mass of CuSO₄ is: 63.55 g/mol + 32.07 g/mol + 64.00 g/mol = 159.62 g/mol.
Important Note: The values used here are approximate. More precise atomic masses can be found in specialized chemical handbooks, but for most practical purposes, the values from a standard periodic table are sufficient.
Significance of Molar Mass in Chemical Calculations
The molar mass of copper(II) sulfate, and any other compound, plays a pivotal role in various chemical calculations:
1. Converting Grams to Moles and Vice Versa:
This is arguably the most frequent application. Knowing the molar mass allows for easy conversion between the mass of a substance (in grams) and the number of moles. For example, if you have 10 grams of CuSO₄, you can calculate the number of moles using the following formula:
Moles = Mass (g) / Molar Mass (g/mol)
Moles of CuSO₄ = 10 g / 159.62 g/mol ≈ 0.0626 moles
Conversely, if you need a specific number of moles, you can calculate the required mass.
2. Stoichiometric Calculations:
Molar mass is fundamental to stoichiometry, the study of quantitative relationships between reactants and products in chemical reactions. Balanced chemical equations use molar ratios to determine the amounts of reactants needed or products formed. The molar mass ensures accurate conversion between moles and mass, leading to precise predictions of reaction yields.
3. Solution Preparation:
Many chemical experiments and industrial processes require preparing solutions of a specific concentration, often expressed as molarity (moles per liter). To prepare a solution of a known molarity, the molar mass of the solute (in this case, CuSO₄) is essential to calculate the required mass to dissolve in a given volume of solvent.
For example, to prepare 1 liter of a 0.1 M CuSO₄ solution, you would need:
Mass = Molarity (mol/L) * Volume (L) * Molar Mass (g/mol)
Mass = 0.1 mol/L * 1 L * 159.62 g/mol = 15.96 g of CuSO₄
4. Determining Empirical and Molecular Formulas:
In analytical chemistry, molar mass can be used in conjunction with elemental analysis data to determine the empirical and molecular formulas of unknown compounds. The empirical formula represents the simplest whole-number ratio of atoms in a compound, while the molecular formula indicates the actual number of atoms. Knowing the molar mass helps distinguish between these two.
Applications of Copper(II) Sulfate
Copper(II) sulfate’s diverse applications stem from its unique properties:
- Agriculture: It acts as a fungicide and algicide, controlling fungal diseases and algae growth in crops and water systems.
- Industry: Used in electroplating, as a mordant in dyeing textiles, and in the production of other copper compounds.
- Medicine: Historically used as an emetic (to induce vomiting) and in some topical treatments; however, its use is now limited due to toxicity concerns.
- Laboratory: A common reagent used in various chemical experiments and analyses, including redox titrations and the preparation of copper-containing solutions.
Hydrates of Copper(II) Sulfate: A Deeper Dive
Copper(II) sulfate readily forms hydrates, meaning it incorporates water molecules into its crystal structure. The most common hydrate is copper(II) sulfate pentahydrate (CuSO₄·5H₂O), a bright blue crystalline solid. The molar mass of the pentahydrate is different from the anhydrous form (CuSO₄).
Calculating the molar mass of CuSO₄·5H₂O:
- CuSO₄: 159.62 g/mol (as calculated previously)
- 5H₂O: 5 * (2 * 1.01 g/mol + 16.00 g/mol) = 90.05 g/mol
Therefore, the molar mass of CuSO₄·5H₂O is: 159.62 g/mol + 90.05 g/mol = 249.67 g/mol.
The presence of water molecules significantly affects the molar mass, and hence, the stoichiometric calculations involving the hydrate. It's crucial to specify whether you are working with the anhydrous or hydrated form of copper(II) sulfate.
Safety Precautions When Handling Copper(II) Sulfate
Copper(II) sulfate, while widely used, is not without its hazards. It's essential to take necessary precautions:
- Eye Protection: Always wear safety goggles to protect your eyes from splashes or dust.
- Skin Protection: Wear gloves to prevent skin contact, as it can cause irritation.
- Ingestion: Avoid ingestion. If ingested, seek immediate medical attention.
- Inhalation: Avoid inhaling dust. Work in a well-ventilated area or use a respirator.
- Disposal: Dispose of waste according to local regulations.
Conclusion: The Importance of Accurate Molar Mass Calculations
The accurate determination and application of the molar mass of copper(II) sulfate are paramount for successful experimentation, precise stoichiometric calculations, and safe handling in diverse applications. Understanding its calculation and the significance of hydrates ensures accurate results and safe laboratory practices. This comprehensive guide has highlighted the fundamental principles, practical applications, and safety considerations associated with this crucial chemical compound. Remember to always use accurate atomic masses from a reliable source and clearly specify whether you are using the anhydrous or hydrated form of the compound in your calculations.
Latest Posts
Latest Posts
-
Liters Per Km To Miles Per Gallon
Apr 05, 2025
-
How Many Tenths Are In A Foot
Apr 05, 2025
-
Write 9 50 As A Decimal Number
Apr 05, 2025
-
What Is 1 4 1 5
Apr 05, 2025
-
Determine Whether The Quantitative Variable Is Discrete Or Continuous
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Molar Mass Of Copper 2 Sulfate . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
