Water Is Made Up Of Which Two Elements
listenit
Apr 04, 2025 · 6 min read
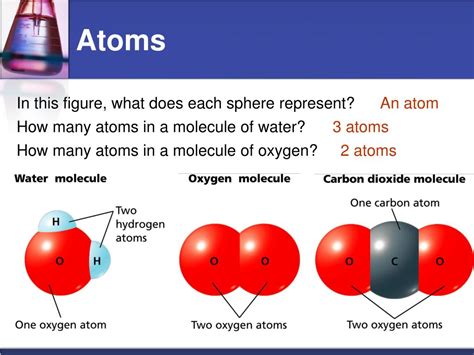
Table of Contents
Water: The Remarkable Compound Made of Two Elements
Water. The very word evokes images of cascading waterfalls, shimmering oceans, and the life-sustaining liquid that flows through our veins. But beyond its aesthetic beauty and biological importance lies a fascinating chemical reality: water is a compound made up of just two elements – hydrogen and oxygen. This seemingly simple combination gives rise to a substance with unique and extraordinary properties that are essential for life as we know it. This article delves deep into the composition of water, exploring its chemical structure, physical properties, and the crucial role it plays in our world.
The Building Blocks: Hydrogen and Oxygen
Before understanding water's composition, let's briefly examine its constituent elements: hydrogen and oxygen.
Hydrogen: The Lightest Element
Hydrogen (H), the first element on the periodic table, is the most abundant element in the universe. It's a highly reactive gas, existing typically as a diatomic molecule (H₂), meaning two hydrogen atoms bonded together. Its single proton and single electron make it incredibly lightweight, contributing to water's relatively low density compared to other liquids. Hydrogen's unique reactivity is key to its role in forming the water molecule.
Oxygen: Essential for Life
Oxygen (O) is the third most abundant element in the universe and a crucial element for life on Earth. It's a highly reactive gas, existing typically as a diatomic molecule (O₂). Oxygen's role in respiration, where it's combined with glucose to release energy, is fundamental to the survival of most organisms. Its strong electronegativity – its tendency to attract electrons – plays a critical role in the polar nature of the water molecule.
The Chemical Structure of Water: H₂O
Water's chemical formula, H₂O, clearly indicates that each molecule of water is comprised of two hydrogen atoms and one oxygen atom. But it's the way these atoms are bonded that gives water its unique characteristics.
Covalent Bonding: Sharing Electrons
The hydrogen and oxygen atoms in a water molecule are connected by covalent bonds. This means that they share electrons to achieve a more stable electron configuration. Oxygen, with six electrons in its outer shell, needs two more to complete its octet (eight electrons). Each hydrogen atom, with one electron, shares its electron with the oxygen atom, forming two covalent bonds. This electron sharing results in a molecule where the oxygen atom is surrounded by eight electrons and each hydrogen atom by two.
Polarity: An Uneven Distribution of Charge
While the covalent bonds are crucial, it's the polarity of the water molecule that truly defines its exceptional properties. Oxygen is significantly more electronegative than hydrogen. This means that the shared electrons spend more time closer to the oxygen atom than to the hydrogen atoms. This unequal sharing creates a slightly negative charge (δ-) on the oxygen atom and slightly positive charges (δ+) on the hydrogen atoms. This makes the water molecule a dipole, possessing a positive and a negative end, like a tiny magnet.
Unique Properties of Water: A Consequence of its Structure
The unique properties of water are a direct consequence of its polar nature and the resulting hydrogen bonding.
Hydrogen Bonding: The Key to Water's Uniqueness
The slightly positive hydrogen atoms of one water molecule are attracted to the slightly negative oxygen atoms of neighboring water molecules. This attraction is called hydrogen bonding. While weaker than covalent bonds, hydrogen bonds are strong enough to significantly influence water's behavior. This extensive network of hydrogen bonds accounts for many of water's remarkable characteristics.
High Boiling Point and Specific Heat Capacity:
Hydrogen bonds are responsible for water's unusually high boiling point (100°C) compared to other molecules of similar size. Breaking these numerous hydrogen bonds requires a significant amount of energy. Similarly, water's high specific heat capacity means it can absorb a large amount of heat with a relatively small temperature change. This property is crucial for regulating temperature in living organisms and maintaining a stable climate on Earth.
Surface Tension and Capillary Action:
The cohesive forces between water molecules due to hydrogen bonding cause high surface tension. This allows water to form droplets and resist external forces. Water's ability to climb against gravity in narrow tubes, known as capillary action, is also a consequence of both cohesion and adhesion (attraction between water molecules and other surfaces). This phenomenon is essential for the transport of water in plants.
Excellent Solvent: The Universal Solvent
Water's polar nature makes it an excellent solvent, often referred to as the "universal solvent." Many ionic compounds and polar molecules dissolve readily in water because their charged particles interact with the charged ends of the water molecules. This ability to dissolve a wide range of substances is crucial for many biological processes, as it allows for the transport of nutrients and waste products within organisms.
Density Anomaly: Ice Floats
Unlike most substances, ice is less dense than liquid water. This unusual property is due to the hydrogen bonding network in ice. The molecules arrange themselves in a crystal lattice structure with more space between them than in liquid water. This lower density means ice floats, preventing bodies of water from freezing solid and allowing aquatic life to survive in winter.
The Importance of Water: Life's Elixir
Water's unique properties are fundamental to life on Earth.
Biological Roles: Solvent, Reactant, and Temperature Regulator
Water acts as a solvent for numerous biochemical reactions, transporting nutrients, removing waste products, and facilitating cell signaling. It's also a crucial reactant in many metabolic processes, such as photosynthesis and respiration. Its high specific heat capacity helps regulate temperature, preventing drastic fluctuations that could damage living organisms.
Ecological Roles: Habitat and Water Cycle
Water is essential for numerous ecosystems, providing habitats for a wide range of organisms. The water cycle, involving evaporation, condensation, and precipitation, continuously recycles water, making it a renewable resource (although its distribution is uneven and its quality can be compromised).
Human Uses: From Drinking to Industry
Water is crucial for human survival, used for drinking, sanitation, agriculture, and numerous industrial processes. Its diverse applications underscore its importance in modern society.
Conclusion: A Remarkable Compound
Water, composed of just two elements, hydrogen and oxygen, possesses remarkable properties that are essential for life and crucial for many natural and human processes. Understanding the chemical structure of water – the covalent bonds, the polarity, and the powerful hydrogen bonds – is key to appreciating its unique characteristics and its indispensable role in our world. From the tiniest cell to the vastest ocean, water's presence shapes our planet and enables the complex web of life that thrives upon it. The seemingly simple formula H₂O hides a profound complexity, a testament to the power of chemistry and the fundamental importance of this extraordinary substance.
Latest Posts
Latest Posts
-
How Many Tenths Are In A Foot
Apr 05, 2025
-
Write 9 50 As A Decimal Number
Apr 05, 2025
-
What Is 1 4 1 5
Apr 05, 2025
-
Determine Whether The Quantitative Variable Is Discrete Or Continuous
Apr 05, 2025
-
How Does Atomic Radius Increase Across The Periodic Table
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Water Is Made Up Of Which Two Elements . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
