How Many Unpaired Electrons Does Manganese Have
listenit
Apr 04, 2025 · 5 min read
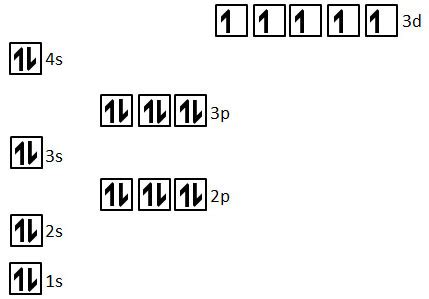
Table of Contents
How Many Unpaired Electrons Does Manganese Have? A Deep Dive into Electronic Configuration and Magnetic Properties
Manganese (Mn), a transition metal residing in the d-block of the periodic table, exhibits fascinating magnetic properties largely dictated by its electronic configuration. Understanding the number of unpaired electrons in manganese is key to comprehending its behavior in various chemical and physical contexts. This comprehensive article will delve into the intricacies of manganese's electronic structure, explaining how to determine the number of unpaired electrons and exploring the implications of this characteristic.
Electronic Configuration: The Foundation of Understanding
The key to determining the number of unpaired electrons lies in understanding manganese's electronic configuration. This configuration describes how electrons are distributed among the various energy levels and sublevels within the atom. Manganese's atomic number is 25, meaning it possesses 25 electrons. Following the Aufbau principle and Hund's rule, these electrons fill the orbitals in a specific order.
Aufbau Principle and Hund's Rule: The Guiding Principles
The Aufbau principle dictates that electrons initially occupy the lowest available energy levels. This means lower energy levels (e.g., 1s, 2s, 2p) are filled before higher energy levels (e.g., 3s, 3p, 4s, 3d).
Hund's rule further specifies that within a subshell (like the 3d subshell), electrons will individually occupy each orbital before pairing up. This maximizes the total spin and results in a more stable configuration.
Manganese's Electronic Configuration: A Step-by-Step Breakdown
Following these principles, the electronic configuration of manganese is: 1s²2s²2p⁶3s²3p⁶4s²3d⁵.
Let's break this down:
- 1s², 2s², 2p⁶, 3s², 3p⁶: These represent the completely filled inner shells, with all electrons paired.
- 4s²: The 4s subshell is also filled with a pair of electrons.
- 3d⁵: This is where the unpaired electrons reside. The 3d subshell has five orbitals, each capable of holding two electrons. According to Hund's rule, each of these five orbitals will receive one electron before any pairing occurs.
Determining the Number of Unpaired Electrons
From the electronic configuration, we can clearly see that the 3d subshell contains five unpaired electrons. Each of the five 3d orbitals holds a single electron, maximizing the total spin and contributing to manganese's magnetic properties. The fully filled inner shells and the filled 4s subshell contribute no unpaired electrons.
The Significance of Unpaired Electrons: Magnetic Properties
The presence of five unpaired electrons in manganese is directly responsible for its paramagnetic behavior. Paramagnetism is a form of magnetism where a material weakly attracts to an external magnetic field. This attraction arises from the interaction between the magnetic moments associated with the unpaired electrons and the external field. Each unpaired electron acts like a tiny magnet, and their combined effect results in a net magnetic moment for the manganese atom.
Contrast with Diamagnetism
In contrast to paramagnetic materials, diamagnetic materials have all their electrons paired. These materials exhibit a very weak repulsion to an external magnetic field. Manganese, due to its unpaired electrons, is decidedly paramagnetic.
Influence on Chemical Reactivity
The unpaired electrons also play a crucial role in manganese's chemical reactivity. These electrons are readily available to participate in chemical bonding, making manganese a relatively reactive metal. Its capacity to exist in multiple oxidation states (+2, +3, +4, +6, +7) is directly related to the number and arrangement of its electrons.
Manganese's Oxidation States and Unpaired Electrons
The oxidation state of manganese refers to the charge it would possess if all its bonds were completely ionic. The different oxidation states arise from the variable loss of electrons from the 4s and 3d orbitals. The number of unpaired electrons can vary depending on the oxidation state:
- Mn²⁺: Loses two electrons, typically from the 4s orbital. This leaves four unpaired electrons in the 3d orbitals.
- Mn³⁺: Loses three electrons, resulting in three unpaired electrons.
- Mn⁴⁺: Loses four electrons, resulting in two unpaired electrons.
- Mn⁷⁺: A highly oxidized state (as in permanganate, MnO₄⁻), this leaves zero unpaired electrons.
Applications Leveraging Manganese's Magnetic Properties
The magnetic properties of manganese, stemming from its unpaired electrons, have numerous applications across various fields:
- Alloys: Manganese is added to steel and other alloys to improve their strength, hardness, and toughness. Its paramagnetic nature contributes to these properties.
- Magnetic Materials: Manganese compounds are incorporated into some magnetic materials, influencing their magnetic behavior.
- Catalysis: Manganese's ability to exist in multiple oxidation states, influenced by its unpaired electrons, makes it a useful catalyst in various chemical reactions. For instance, manganese oxides are used as catalysts in the production of certain chemicals.
- Biological Systems: Manganese plays a vital role in biological systems, acting as a cofactor in several enzymes. Its ability to accept and donate electrons is crucial for its biological function.
Conclusion: The Importance of Electronic Configuration
The number of unpaired electrons in manganese, directly determined by its electronic configuration, is a fundamental characteristic that significantly impacts its physical and chemical properties. Its paramagnetism, variable oxidation states, and reactivity are all consequences of this key feature. Understanding the electronic configuration and the resultant unpaired electrons is crucial for appreciating the unique role manganese plays in diverse applications, from metallurgy to biology. Further exploration into the specifics of manganese's interactions in various compounds and environments will continue to reveal new and exciting applications for this versatile transition metal. Further research continually unveils deeper understanding of the implications of this seemingly simple concept – the number of unpaired electrons in manganese. This continues to drive innovation in materials science, chemistry, and biological research.
Latest Posts
Latest Posts
-
Liters Per Km To Miles Per Gallon
Apr 05, 2025
-
How Many Tenths Are In A Foot
Apr 05, 2025
-
Write 9 50 As A Decimal Number
Apr 05, 2025
-
What Is 1 4 1 5
Apr 05, 2025
-
Determine Whether The Quantitative Variable Is Discrete Or Continuous
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about How Many Unpaired Electrons Does Manganese Have . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
