Dissolution Of Sodium Chloride In Water
listenit
Apr 03, 2025 · 6 min read
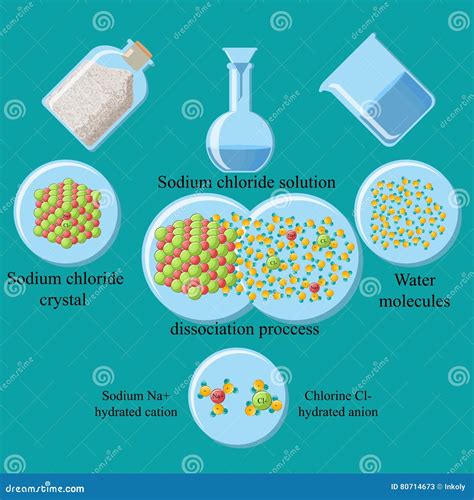
Table of Contents
Dissolution of Sodium Chloride in Water: A Deep Dive
The seemingly simple act of dissolving table salt (sodium chloride, NaCl) in water is a fascinating example of chemistry in action. It's a process we encounter daily, yet the underlying mechanisms and factors influencing it are surprisingly complex. This article will explore the dissolution of sodium chloride in water in detail, covering the key concepts, influencing factors, and applications of this fundamental process.
Understanding the Basics: Polarity and Ion-Dipole Interactions
The key to understanding why sodium chloride dissolves readily in water lies in the properties of both substances. Water (H₂O) is a polar molecule, meaning it possesses a partial positive charge on the hydrogen atoms and a partial negative charge on the oxygen atom. This polarity arises from the difference in electronegativity between oxygen and hydrogen. Sodium chloride, on the other hand, is an ionic compound, formed by the electrostatic attraction between positively charged sodium ions (Na⁺) and negatively charged chloride ions (Cl⁻).
When sodium chloride is added to water, the polar water molecules interact with the charged ions. This interaction is known as ion-dipole interaction. The partially negative oxygen atoms of water molecules are attracted to the positively charged sodium ions, while the partially positive hydrogen atoms are attracted to the negatively charged chloride ions.
The Hydration Process: Surrounding Ions with Water Molecules
These ion-dipole interactions are strong enough to overcome the electrostatic forces holding the sodium and chloride ions together in the crystal lattice of the solid NaCl. This process is called hydration, where water molecules surround each ion, forming a hydration shell. Each ion becomes surrounded by a sphere of water molecules, effectively shielding it from the other ions and preventing the reformation of the crystal lattice.
The energy released during the hydration process is significant and helps to drive the dissolution process forward. This energy is known as the heat of hydration. It's important to note that while dissolving salt in water is often perceived as a cooling process, the actual dissolution process is exothermic (releases heat). The perceived cooling effect is often due to the heat absorbed from the surroundings as the salt dissolves, overcoming the higher enthalpy required for lattice disruption.
Factors Affecting the Dissolution Rate of Sodium Chloride
Several factors can influence the rate at which sodium chloride dissolves in water:
1. Temperature: The Heat Factor
Increasing the temperature of the water generally increases the dissolution rate of sodium chloride. Higher temperatures provide water molecules with greater kinetic energy, leading to more frequent and forceful collisions with the NaCl crystal lattice. This increased energy facilitates the breaking of ionic bonds and accelerates the hydration process.
2. Surface Area: More Contact, Faster Dissolution
The surface area of the NaCl crystal exposed to the water also plays a crucial role. Crushing or grinding the salt into a finer powder significantly increases its surface area, providing more points of contact for water molecules to interact with. This leads to a much faster dissolution rate.
3. Agitation: Stirring it Up
Stirring or agitating the solution helps to remove the hydrated ions from the vicinity of the NaCl crystal surface. This prevents the formation of a saturated layer around the crystal, which would otherwise hinder further dissolution. By constantly replenishing the water surrounding the crystals with fresh, un-saturated water, the process is accelerated.
4. Concentration: Saturation Point
The concentration of the solution also affects the dissolution rate. As more NaCl dissolves, the concentration of Na⁺ and Cl⁻ ions in the solution increases. Eventually, the solution becomes saturated, meaning it cannot dissolve any more NaCl at that temperature. At saturation, the rate of dissolution equals the rate of recrystallization, resulting in no net change in the amount of dissolved salt.
Beyond Simple Dissolution: Understanding Solubility and Equilibrium
The dissolution of sodium chloride in water is a dynamic equilibrium. Even in a saturated solution, the process of dissolution and recrystallization continues at equal rates. This is described by the solubility product constant (Ksp), which represents the equilibrium constant for the dissolution reaction:
NaCl(s) ⇌ Na⁺(aq) + Cl⁻(aq)
Ksp = [Na⁺][Cl⁻]
The value of Ksp is temperature-dependent, reflecting the fact that solubility changes with temperature. For NaCl, the solubility is relatively high and increases only moderately with temperature.
Applications of Sodium Chloride Dissolution
The dissolution of sodium chloride in water is a fundamental process with a wide range of applications, including:
1. Electrolyte Solutions: Conductivity and Charge Transfer
Dissolving NaCl in water creates an electrolyte solution, meaning it can conduct electricity. The presence of freely moving ions (Na⁺ and Cl⁻) allows the flow of electrical current. This property is crucial in various applications, including batteries, fuel cells, and electroplating.
2. Biological Systems: Essential for Life
Sodium chloride plays a vital role in many biological systems. The proper balance of sodium and chloride ions is essential for maintaining osmotic pressure, nerve impulse transmission, and muscle contraction. The dissolution of NaCl in bodily fluids ensures the availability of these ions for these crucial functions.
3. Industrial Processes: Cleaning, Food Preservation, and More
Dissolution of NaCl is used extensively in various industrial processes. It's utilized in food preservation (pickling, salting), cleaning processes, water treatment (to adjust salinity and aid in disinfection), and in the production of various chemicals.
4. Chemical Reactions: A Versatile Reactant and Solvent
Dissolved NaCl is employed in numerous chemical reactions as a source of sodium and chloride ions. Its role in these reactions can vary from providing a specific ion for reaction to helping control solution properties like ionic strength. Furthermore, saturated NaCl solutions can act as a convenient way to extract or precipitate certain compounds through salting-out effects.
Advanced Considerations: Factors Beyond the Basics
While the basic principles outlined above provide a good understanding of NaCl dissolution, several more nuanced aspects require consideration:
1. Activity Coefficients: Deviations from Ideality
In concentrated solutions, the interactions between ions become significant, leading to deviations from ideal behavior. Activity coefficients are used to correct for these deviations and accurately reflect the effective concentration of ions in the solution.
2. The Role of Impurities: Affecting Solubility
Impurities in the NaCl crystal or the water can significantly influence the dissolution rate and solubility. These impurities can interfere with the hydration process or alter the crystal structure, affecting the ease with which ions are released.
3. Common Ion Effect: Suppressing Solubility
The presence of a common ion can suppress the solubility of NaCl. If another soluble chloride salt is added to a NaCl solution, the increase in Cl⁻ concentration shifts the equilibrium of the NaCl dissolution reaction to the left, reducing the solubility of NaCl.
Conclusion: A Simple Process with Profound Implications
The dissolution of sodium chloride in water is a seemingly simple process, yet it illustrates fundamental principles of chemistry and has far-reaching implications in various fields. Understanding the factors affecting dissolution rate and equilibrium is essential in many scientific and industrial applications. From maintaining the proper electrolyte balance in our bodies to enabling numerous industrial processes, the seemingly simple act of dissolving salt in water plays a surprisingly significant role in our world. This detailed exploration should provide a solid foundation for further investigation into this ubiquitous process.
Latest Posts
Latest Posts
-
How To Find The Slope Of Demand Curve
Apr 03, 2025
-
How Did He Know That The Nucleus Was Positively Charged
Apr 03, 2025
-
What Is 4 To The 5 Power
Apr 03, 2025
-
Integral 1 X 2 3 2
Apr 03, 2025
-
Y 3 X 5 X 2
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Dissolution Of Sodium Chloride In Water . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
