How Did He Know That The Nucleus Was Positively Charged
listenit
Apr 03, 2025 · 6 min read
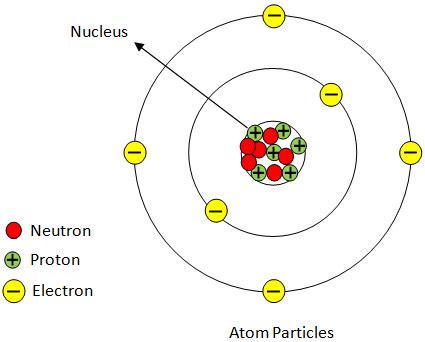
Table of Contents
How Did He Know That the Nucleus Was Positively Charged? Unraveling the Discovery of the Atomic Nucleus
The discovery that the atomic nucleus is positively charged wasn't a singular "eureka!" moment, but rather a culmination of ingenious experiments, insightful interpretations, and the relentless pursuit of understanding the fundamental building blocks of matter. This journey involved numerous scientists, building upon each other's work, gradually painting a clearer picture of the atom's structure. This article delves into the pivotal experiments and reasoning that led to this groundbreaking conclusion, highlighting the contributions of key figures like Ernest Rutherford and his predecessors.
The Pre-Nuclear Atom: A Shifting Landscape of Understanding
Before we delve into the discovery of the positively charged nucleus, it's crucial to understand the prevailing atomic models that existed beforehand. These models, while ultimately inaccurate, provided the crucial stepping stones for the revolutionary discoveries to come.
Thomson's Plum Pudding Model: A Sweet, But Wrong, Analogy
J.J. Thomson's discovery of the electron in 1897 dramatically changed the understanding of the atom. His experiments with cathode rays revealed the existence of negatively charged particles, far smaller than the atom itself. This led him to propose the "plum pudding" model: a positively charged sphere (the pudding) with negatively charged electrons (the plums) embedded within it. This model, while a significant advancement, was fundamentally flawed. It couldn't explain the stability of the atom, nor could it account for the results of future experiments.
The Need for a New Model: Experimental Inconsistencies
The plum pudding model faced significant challenges. The lack of a defined structure within the atom couldn't explain various observations, particularly those involving the scattering of alpha particles. Alpha particles, positively charged particles emitted by certain radioactive substances, were becoming a crucial tool in probing the structure of matter. The way these alpha particles interacted with matter would provide the key evidence to overturn Thomson's model.
Rutherford's Gold Foil Experiment: A Revolutionary Breakthrough
Ernest Rutherford, along with his team members Hans Geiger and Ernest Marsden, conducted a pivotal experiment in 1909 that shattered the plum pudding model. This experiment, famously known as the gold foil experiment, involved bombarding a thin gold foil with a beam of alpha particles. Based on the plum pudding model, they expected the alpha particles to pass through the foil with only minor deflections. The results were astonishing.
Unexpected Deflections: Challenging the Plum Pudding Model
Most of the alpha particles did indeed pass through the gold foil undeflected, as expected. However, a small but significant fraction of the alpha particles were deflected at large angles, some even bouncing straight back! This unexpected result was completely inconsistent with Thomson's model. If the positive charge were uniformly distributed, as the plum pudding model suggested, the alpha particles should have experienced only minor deflections.
The Birth of the Nuclear Model: Interpreting the Results
Rutherford's genius lay in his interpretation of these unexpected results. He reasoned that the large-angle scattering could only be explained if the positive charge within the atom was concentrated in a tiny, dense region at the center – the nucleus. This nucleus would contain most of the atom's mass and its positive charge. The electrons, being much lighter and negatively charged, would orbit this central nucleus at a significant distance.
The Positively Charged Nucleus: Evidence and Reasoning
The conclusion that the nucleus is positively charged wasn't solely based on the deflection of alpha particles. Several lines of evidence and reasoning supported this revolutionary claim:
1. The Magnitude of Deflection: Quantifying the Force
The angle of deflection of the alpha particles was directly related to the strength of the repulsive force between the positively charged alpha particle and the positive charge within the atom. The larger the deflection, the stronger the repulsive force, indicating a concentrated positive charge. Rutherford's team meticulously measured these deflections, providing quantitative support for his nuclear model.
2. The Rarity of Large-Angle Deflections: Size of the Nucleus
The fact that only a small fraction of alpha particles experienced large-angle deflections suggested that the positively charged region was incredibly small compared to the overall size of the atom. Most alpha particles passed through the mostly empty space surrounding the nucleus, encountering only minimal electrostatic interaction. This implied that the atom was predominantly empty space, a radical departure from previous models.
3. The Straight-Back Scattering: Direct Collision
The most striking evidence was the observation of alpha particles bouncing straight back. This could only occur if an alpha particle experienced a head-on collision with the dense, positively charged nucleus. The repulsive force from such a close encounter was strong enough to reverse the direction of the alpha particle's motion.
4. Radioactivity: Further Confirmation
The discovery and study of radioactivity played a crucial role. The alpha particles themselves, being positively charged, provided a natural probe to investigate the structure of matter. The behavior of other radioactive emissions, like beta particles (negatively charged) and gamma rays (neutral), also contributed to a more complete picture of the atom, indirectly supporting the concept of a positively charged nucleus.
Beyond Rutherford: Refinements and Further Discoveries
Rutherford's nuclear model was a monumental leap forward, but it was not without its limitations. It couldn't explain the stability of the atom – why the orbiting electrons didn't collapse into the nucleus due to electromagnetic attraction. This issue would later be addressed by Niels Bohr's model, incorporating quantum mechanics.
The subsequent development of quantum mechanics further refined our understanding of the atom's structure, leading to the modern quantum mechanical model. This model, while significantly more complex than Rutherford's model, retains the fundamental concept of a positively charged nucleus surrounded by orbiting electrons.
Conclusion: A Legacy of Discovery
The discovery of the positively charged nucleus was a pivotal moment in the history of science. It fundamentally changed our understanding of the atom, opening the door for countless advancements in physics, chemistry, and related fields. It stands as a testament to the power of careful experimentation, insightful interpretation, and the collaborative spirit of scientific inquiry. Rutherford's gold foil experiment, and the insightful analysis that followed, remain a shining example of how scientific breakthroughs can transform our understanding of the universe at its most fundamental level. The positively charged nucleus is not just a scientific fact; it is a cornerstone of modern science, a concept that continues to shape our understanding of matter and energy. The legacy of this discovery continues to inspire generations of scientists, reminding us of the power of curiosity and the relentless pursuit of knowledge.
Latest Posts
Latest Posts
-
How Long To Travel 4 Light Years
Apr 04, 2025
-
What Is The Value Of 5
Apr 04, 2025
-
Two Strands Of Dna Are Held Together By
Apr 04, 2025
-
The Muscle That Subdivides The Ventral Body Cavity
Apr 04, 2025
-
Boron Is Solid Liquid Or Gas
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about How Did He Know That The Nucleus Was Positively Charged . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
