A Solution In Which The Solvent Is Water
listenit
Apr 02, 2025 · 7 min read
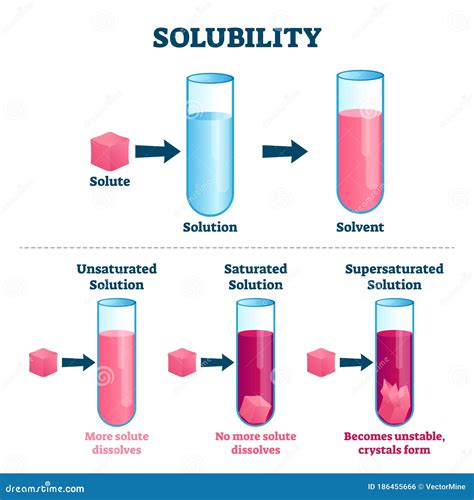
Table of Contents
Aqueous Solutions: A Deep Dive into the Chemistry of Water as a Solvent
Water, often taken for granted as the elixir of life, is far more than just a simple molecule (H₂O). Its unique properties make it an exceptional solvent, capable of dissolving a vast array of substances and playing a crucial role in countless chemical and biological processes. This article explores the fascinating world of aqueous solutions, delving into the factors that govern solubility, the types of solutions formed, and the implications of these solutions across various fields.
Understanding the Nature of Water as a Solvent
Water's unparalleled solvency stems from its polarity. The oxygen atom, being more electronegative than the hydrogen atoms, attracts the shared electrons more strongly, creating a partial negative charge (δ-) on the oxygen and partial positive charges (δ+) on the hydrogens. This creates a dipole moment, essentially making the water molecule a tiny magnet with a positive and negative end.
The Role of Hydrogen Bonding
This polarity is further amplified by hydrogen bonding. The partially positive hydrogen atom of one water molecule is strongly attracted to the partially negative oxygen atom of another water molecule. This relatively strong intermolecular force creates a cohesive network of water molecules, influencing many of its properties, including its high boiling point, surface tension, and exceptional ability to dissolve polar and ionic substances.
Dissolving Ionic Compounds
When an ionic compound, like sodium chloride (NaCl), is added to water, the polar water molecules surround the ions. The partially negative oxygen atoms are attracted to the positively charged sodium ions (Na⁺), while the partially positive hydrogen atoms are attracted to the negatively charged chloride ions (Cl⁻). This process, known as hydration, weakens the electrostatic forces holding the ions together in the crystal lattice, eventually separating them and allowing them to disperse throughout the solution. This results in a homogeneous mixture where the ions are surrounded by a hydration shell of water molecules.
Dissolving Polar Covalent Compounds
Polar covalent compounds, such as sugars and alcohols, also readily dissolve in water. The polar water molecules interact with the polar functional groups (like hydroxyl –OH groups) of these molecules through dipole-dipole interactions and hydrogen bonding. These interactions help to overcome the intermolecular forces within the solute, allowing it to dissolve and disperse uniformly in the water.
Factors Affecting Solubility in Aqueous Solutions
Several factors influence the solubility of a substance in water:
1. Temperature
Generally, the solubility of most solids in water increases with increasing temperature. As the temperature rises, the kinetic energy of the water molecules increases, allowing them to more effectively overcome the intermolecular forces holding the solute particles together. However, the solubility of gases in water typically decreases with increasing temperature. This is because higher temperatures increase the kinetic energy of gas molecules, allowing them to escape the liquid phase more easily.
2. Pressure
Pressure significantly affects the solubility of gases in water, but has a negligible effect on the solubility of solids and liquids. According to Henry's Law, the solubility of a gas is directly proportional to the partial pressure of that gas above the liquid. Increasing the pressure above the solution forces more gas molecules into the liquid phase, increasing its solubility.
3. Polarity
The principle of "like dissolves like" governs solubility. Polar solvents, like water, dissolve polar and ionic solutes effectively. Nonpolar substances, however, tend to be insoluble in water because they lack the ability to form strong interactions with water molecules. Instead, they tend to clump together, minimizing their contact with the water.
4. Molecular Size and Structure
Larger molecules generally have lower solubility than smaller molecules. The increased surface area of larger molecules may increase the number of interactions with water but the strength of these interactions may not be sufficient to overcome the stronger intermolecular forces within the larger molecule. The specific arrangement of atoms within a molecule (its structure) also plays a crucial role in determining its solubility. The presence of functional groups that can participate in hydrogen bonding or other strong interactions enhances solubility.
Types of Aqueous Solutions
Aqueous solutions can be classified in several ways:
1. Based on the amount of solute
- Dilute solutions: Contain a relatively small amount of solute compared to the solvent.
- Concentrated solutions: Contain a relatively large amount of solute compared to the solvent. The exact definition of "dilute" and "concentrated" is context-dependent.
2. Based on the ability to dissolve more solute
- Unsaturated solutions: Can dissolve more solute at a given temperature and pressure.
- Saturated solutions: Contain the maximum amount of solute that can dissolve at a given temperature and pressure. Any additional solute will remain undissolved.
- Supersaturated solutions: Contain more solute than the saturation point at a given temperature and pressure. These are unstable and tend to precipitate out excess solute if disturbed.
3. Based on the conductivity of electricity
- Electrolyte solutions: Contain dissolved substances that conduct electricity. These typically include ionic compounds which dissociate into ions when dissolved in water. Strong electrolytes completely dissociate, while weak electrolytes only partially dissociate.
- Nonelectrolyte solutions: Contain dissolved substances that do not conduct electricity. These are typically covalent compounds that do not dissociate into ions.
Applications of Aqueous Solutions
The ubiquitous nature of aqueous solutions highlights their significance across numerous disciplines:
1. Biological Systems
Water is the primary solvent in all living organisms. Biological processes, from metabolism to cellular transport, rely heavily on aqueous solutions. Blood, for example, is an aqueous solution containing various dissolved substances like ions, proteins, and gases, essential for transporting nutrients and oxygen throughout the body.
2. Industrial Processes
Many industrial processes use aqueous solutions. The chemical industry utilizes them extensively in reactions, extractions, and purifications. Aqueous solutions are also vital in cleaning processes, food processing, and the production of many consumer goods.
3. Environmental Science
Understanding aqueous solutions is crucial for environmental management. The behavior of pollutants in water bodies, the solubility of nutrients in soil, and the dynamics of aquatic ecosystems are all influenced by the properties of aqueous solutions. Pollution control and water treatment strategies often rely on manipulating the solubility and reactivity of substances in water.
4. Pharmaceutical Sciences
Many pharmaceuticals are administered as aqueous solutions or suspensions. Understanding the solubility and stability of drugs in water is crucial for developing effective drug delivery systems. The formulation of medications often involves careful control of the pH and other factors to ensure the desired solubility and bioavailability of the active ingredients.
Analyzing Aqueous Solutions: Concentration Units
Describing the composition of an aqueous solution accurately requires understanding various concentration units. These units express the amount of solute present relative to the amount of solvent or solution. Common concentration units include:
- Molarity (M): Moles of solute per liter of solution. This is a widely used unit in chemistry and biochemistry.
- Molality (m): Moles of solute per kilogram of solvent. Molality is temperature-independent, unlike molarity, which can change with temperature due to volume changes.
- Mass percent (% w/w): Grams of solute per 100 grams of solution.
- Volume percent (% v/v): Milliliters of solute per 100 milliliters of solution. This is typically used for liquid-liquid solutions.
- Parts per million (ppm) and parts per billion (ppb): These units are used to express very low concentrations, typically for pollutants or trace elements.
Conclusion
Aqueous solutions are fundamental to chemistry, biology, and numerous other scientific disciplines. Understanding their properties, the factors influencing solubility, and the various types of solutions is essential for interpreting the world around us and developing technologies that improve our lives. From the intricate processes within our cells to large-scale industrial applications, water's remarkable solvency continues to shape our world in profound ways. The continued investigation and innovative applications of aqueous solutions promise to yield even more remarkable advancements in the future.
Latest Posts
Latest Posts
-
What Is The Charge Of Chlorine
Apr 03, 2025
-
Area Of A Circle With A Radius Of 7
Apr 03, 2025
-
Find The Slope Of The Line Perpendicular
Apr 03, 2025
-
What Is The Square Root Of 59
Apr 03, 2025
-
What Is The Square Root Of 147
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about A Solution In Which The Solvent Is Water . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
