What Is The Charge Of Chlorine
listenit
Apr 03, 2025 · 5 min read
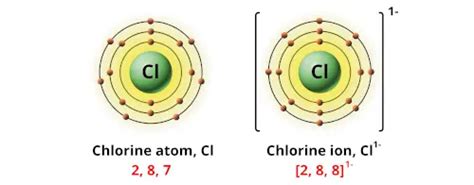
Table of Contents
What is the Charge of Chlorine? Understanding Chlorine's Oxidation States and Reactivity
Chlorine, a vibrant yellow-green gas, is a fascinating element with a rich history and diverse applications. From its crucial role in purifying drinking water to its use in various industrial processes, chlorine's impact is undeniable. However, understanding its behavior requires a deeper dive into its chemical properties, particularly its charge or, more accurately, its oxidation states. This article will comprehensively explore the charge of chlorine, delving into its varying oxidation states, how these states affect its reactivity, and the implications of this reactivity in different contexts.
Understanding Oxidation States: Beyond Simple Charges
Before we delve into the specifics of chlorine's charge, it's crucial to clarify the concept of oxidation states. While often simplified as "charge," the oxidation state represents the hypothetical charge an atom would have if all bonds to atoms of different elements were 100% ionic. This is a crucial distinction because chlorine, unlike many ionic species, often forms covalent bonds where electrons are shared, not fully transferred. Therefore, speaking of chlorine's charge is an oversimplification. Its oxidation state, however, provides a more accurate representation of its electron distribution in a molecule or compound.
Chlorine, being in group 17 (halogens) of the periodic table, possesses seven valence electrons. This means it has a strong tendency to gain one electron to achieve a stable octet configuration, resembling the noble gas argon. This tendency dictates its most common oxidation state.
The Most Common Oxidation State: -1
The most prevalent oxidation state of chlorine is -1. This arises when chlorine gains one electron, fulfilling its desire for a stable octet. This is observed in numerous chlorine-containing compounds:
-
Chlorides: In simple chlorides like sodium chloride (NaCl), potassium chloride (KCl), and hydrogen chloride (HCl), chlorine exists in a -1 oxidation state. The electron is effectively transferred from the more electropositive metal or hydrogen atom to the chlorine atom.
-
Organic Chlorides: Many organic molecules incorporate chlorine atoms. In these compounds, chlorine typically exhibits a -1 oxidation state, forming a covalent bond with a carbon atom. Examples include chloroform (CHCl₃) and dichloromethane (CH₂Cl₂).
-
Other Covalent Compounds: Even in covalent compounds where chlorine is bonded to other nonmetals, it often retains a -1 oxidation state. This is because chlorine is more electronegative than many of these elements, effectively attracting the shared electrons closer to itself.
Chlorine's Variable Oxidation States: Beyond -1
While -1 is the most common, chlorine's versatility extends to a range of oxidation states, showcasing its remarkable reactivity. The following are some other significant oxidation states:
-
0: Chlorine exists in the elemental state (Cl₂) with an oxidation state of 0. In this form, chlorine atoms share electrons equally in a covalent bond.
-
+1: This oxidation state is less common but appears in compounds such as hypochlorous acid (HOCl), a powerful disinfectant.
-
+3: Chlorous acid (HClO₂) displays chlorine in the +3 oxidation state.
-
+5: Chloric acid (HClO₃) represents chlorine in the +5 oxidation state. Chlorates, salts of chloric acid, find applications in various industries.
-
+7: Perchloric acid (HClO₄) showcases the highest common oxidation state of chlorine, +7. Perchlorates, the salts of perchloric acid, are powerful oxidizing agents.
The Impact of Oxidation State on Reactivity
The diverse oxidation states of chlorine directly influence its reactivity. The -1 oxidation state represents a relatively stable form, as it achieves a full octet. However, chlorine in higher oxidation states (+1, +3, +5, +7) possesses a strong oxidizing ability. This means it readily accepts electrons from other substances, leading to redox reactions. These reactions can be harnessed for various applications, including:
-
Disinfection: Hypochlorous acid (HOCl), with chlorine in the +1 oxidation state, is a potent disinfectant, widely used in water treatment and sanitation. Its oxidizing ability disrupts the cellular processes of bacteria and other pathogens.
-
Bleaching: The oxidizing power of chlorine in higher oxidation states is utilized in bleaching agents. These agents oxidize colored compounds, leading to their discoloration.
-
Industrial Processes: Chlorine's oxidizing capabilities are crucial in various industrial processes, including the manufacturing of certain plastics and pharmaceuticals.
-
Rocket Propellants: Certain chlorine-containing compounds with chlorine in high oxidation states are used as components of rocket propellants due to their high energy content.
Factors Affecting Chlorine's Oxidation State
Several factors influence the oxidation state of chlorine in a particular compound:
-
Electronegativity: Chlorine's relatively high electronegativity leads it to attract electrons towards itself, often resulting in a negative oxidation state (-1). However, when it bonds with more electronegative elements like oxygen, it can exhibit positive oxidation states.
-
Bonding Partners: The nature of the atoms it bonds with significantly impacts chlorine's oxidation state. Bonds with electropositive metals typically result in -1, whereas bonds with more electronegative elements like oxygen lead to positive oxidation states.
-
Reaction Conditions: The reaction conditions, such as temperature and pressure, can also influence the oxidation state of chlorine in the products formed.
Safety Considerations: Handling Chlorine and its Compounds
It's essential to emphasize the importance of safe handling practices when dealing with chlorine and its compounds. Chlorine gas is toxic and corrosive, requiring appropriate safety precautions, including proper ventilation and protective equipment. Many chlorine compounds are also hazardous, and their handling should adhere to relevant safety regulations and guidelines.
Conclusion: A Versatile Element with Diverse Applications
Chlorine's versatility, stemming directly from its various oxidation states, makes it a crucial element in numerous applications. While its most common oxidation state is -1, the ability to exist in higher oxidation states (+1, +3, +5, +7) significantly enhances its reactivity and opens the door to a diverse range of uses, from water purification to industrial processes. Understanding these oxidation states and their impact on reactivity is crucial for appreciating chlorine's significant role in the world around us. However, always remember that safe handling practices are paramount when working with this potent element and its compounds. Remember to always consult safety data sheets (SDS) before handling any chemicals.
Latest Posts
Latest Posts
-
Lewis Structure For N Ch3 3
Apr 04, 2025
-
How Many Feet Is A Marathon
Apr 04, 2025
-
Substance That Is Dissolved In A Solution
Apr 04, 2025
-
Is Air A Compound Or Element Or Mixture
Apr 04, 2025
-
What Organelle Is Missing From The Red Blood Cells
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about What Is The Charge Of Chlorine . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
