Substance That Is Dissolved In A Solution
listenit
Apr 04, 2025 · 6 min read
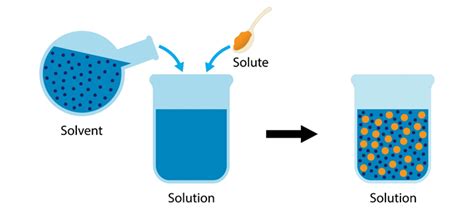
Table of Contents
The Fascinating World of Solutes: Understanding Substances Dissolved in Solutions
Chemistry, at its core, is the study of matter and its transformations. A fundamental concept within chemistry is the solution, a homogeneous mixture composed of two or more substances. Understanding solutions requires understanding their components: the solvent, which dissolves the other substance, and the solute, the substance being dissolved. This article delves deep into the world of solutes, exploring their properties, behavior in solutions, and their significance across various scientific disciplines.
What is a Solute? A Deeper Dive
A solute is any substance that dissolves in a solvent to form a solution. It can be a solid, liquid, or gas. The key characteristic of a solute is its ability to disperse uniformly throughout the solvent at a molecular or ionic level, resulting in a homogeneous mixture where the solute is no longer visibly distinct. This process is known as dissolution.
The amount of solute that can dissolve in a given amount of solvent at a specific temperature and pressure is known as its solubility. Solubility is a crucial property that dictates the concentration of a solution. Factors influencing solubility include the nature of the solute and solvent (like dissolves like), temperature, and pressure.
Types of Solutes: A Categorical Overview
Solutes can be categorized in several ways:
-
By chemical nature: Solutes can be ionic compounds (e.g., NaCl), covalent compounds (e.g., sugar), or metallic compounds. Ionic compounds dissociate into ions when dissolved, while covalent compounds typically remain intact as molecules.
-
By polarity: Solutes can be polar (having a significant difference in electronegativity between atoms) or nonpolar (having little to no difference in electronegativity). Polar solutes generally dissolve well in polar solvents (like water), while nonpolar solutes dissolve well in nonpolar solvents (like oil). This is often summarized by the adage: "like dissolves like."
-
By size: The size of solute molecules or ions affects their solubility. Smaller particles tend to dissolve more readily than larger ones.
-
By functionality: In biological systems, solutes can be categorized by their function, such as electrolytes (ions that conduct electricity), nutrients, or waste products.
The Dissolution Process: A Molecular Perspective
The dissolution process is a dynamic equilibrium between the solute dissolving and the solute precipitating out of solution. Several factors influence the rate of dissolution:
-
Surface area: Increasing the surface area of the solute (e.g., by grinding a solid into a powder) increases the rate of dissolution. More solute particles are exposed to the solvent, increasing the chances of interaction and dissolution.
-
Agitation: Stirring or shaking a solution increases the rate of dissolution by bringing fresh solvent into contact with the solute particles.
-
Temperature: Increasing the temperature generally increases the rate of dissolution for solids and liquids in liquids. Higher temperatures provide the solute particles with more kinetic energy, making it easier for them to overcome intermolecular forces and dissolve. The effect of temperature on the dissolution of gases is the opposite; increasing temperature decreases the solubility of gases.
-
Pressure: Pressure has a significant effect on the solubility of gases in liquids. Increasing the pressure increases the solubility of gases, according to Henry's Law. The effect of pressure on the solubility of solids and liquids is generally negligible.
Understanding Intermolecular Forces
The dissolution process is fundamentally driven by intermolecular forces. When a solute dissolves in a solvent, the intermolecular forces between solute particles and solvent particles must overcome the intermolecular forces within the solute and within the solvent.
-
Solute-solvent interactions: Strong solute-solvent interactions (e.g., hydrogen bonding, dipole-dipole interactions, ion-dipole interactions) facilitate dissolution. The solvent molecules surround the solute particles, effectively shielding them from each other and preventing them from aggregating.
-
Solute-solute interactions: Strong solute-solute interactions (e.g., strong ionic bonds, strong hydrogen bonds) hinder dissolution. Energy is required to overcome these interactions before the solute particles can be separated and surrounded by solvent molecules.
-
Solvent-solvent interactions: Solvent-solvent interactions also play a role. Breaking these interactions requires energy, which can affect the overall dissolution process.
Concentration of Solutions: Expressing the Amount of Solute
The concentration of a solution describes the amount of solute present in a given amount of solution or solvent. Several methods exist for expressing concentration, each with its advantages and disadvantages:
-
Molarity (M): Molarity is defined as the number of moles of solute per liter of solution. It is widely used in chemistry due to its direct relationship with stoichiometry.
-
Molality (m): Molality is defined as the number of moles of solute per kilogram of solvent. Unlike molarity, molality is independent of temperature, making it useful in situations where temperature changes significantly.
-
Mass percentage (% w/w): Mass percentage is the mass of solute divided by the total mass of the solution, multiplied by 100%.
-
Volume percentage (% v/v): Volume percentage is the volume of solute divided by the total volume of the solution, multiplied by 100%.
-
Parts per million (ppm) and parts per billion (ppb): These units are used to express very low concentrations of solutes.
The Significance of Solutes Across Disciplines
The concept of solutes and their behavior in solutions is fundamental across numerous scientific fields:
-
Biology: Biological systems rely heavily on solutions. Blood, for instance, is a complex solution containing various solutes like glucose, ions, and proteins. Understanding solute concentrations is crucial for maintaining homeostasis and overall health.
-
Medicine: Many drugs are administered as solutions, allowing for controlled drug delivery and better absorption by the body. Intravenous solutions deliver essential nutrients and electrolytes directly into the bloodstream.
-
Environmental science: Water quality is largely determined by the types and concentrations of solutes present. Understanding solute behavior is crucial for monitoring pollution and protecting water resources.
-
Geology: The dissolution of minerals in groundwater plays a significant role in shaping landscapes and forming caves. Understanding solute transport in geological systems is critical for managing groundwater resources.
-
Chemistry: Solutions are essential in numerous chemical reactions. The concentration of reactants and products in solution dictates the rate and equilibrium of these reactions.
Conclusion: The Ubiquity of Solutes
Solutes are integral components of solutions, their presence dictating numerous properties and behaviors. Their dissolution process, governed by intermolecular forces and influenced by various factors, is a complex yet fascinating aspect of chemistry. Understanding solutes is crucial across various disciplines, highlighting their importance in numerous natural and engineered systems. From biological processes to environmental monitoring to industrial applications, the world of solutes is extensive and continues to be a vibrant area of scientific inquiry. Further research continues to reveal the intricacies of solute behavior, leading to new discoveries and applications in various fields.
Latest Posts
Latest Posts
-
Rectangle That Is Not A Parallelogram
Apr 04, 2025
-
How Many Electrons Does Carbon Have Available For Chemical Bonding
Apr 04, 2025
-
What Is It Called When Everyone Plays Together
Apr 04, 2025
-
Y Is Inversely Proportional To X
Apr 04, 2025
-
Is Baking Bread A Chemical Or Physical Change
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Substance That Is Dissolved In A Solution . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
