How Many Electrons Does Carbon Have Available For Chemical Bonding
listenit
Apr 04, 2025 · 5 min read
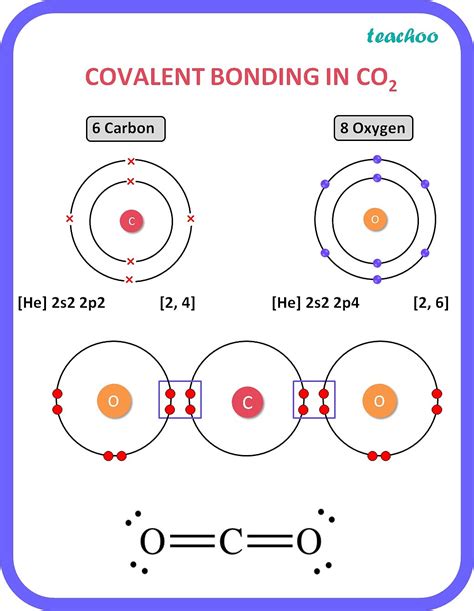
Table of Contents
How Many Electrons Does Carbon Have Available for Chemical Bonding?
Carbon, the cornerstone of organic chemistry and the building block of life, possesses a unique electronic configuration that allows it to form an incredibly diverse range of molecules. Understanding how many electrons carbon has available for chemical bonding is crucial to comprehending its remarkable versatility and the vast complexity of organic chemistry. This article delves deep into the electronic structure of carbon, explaining its bonding capabilities, and exploring the implications for its prevalence in biological and material sciences.
Carbon's Electronic Structure: The Key to its Bonding
Carbon's atomic number is 6, meaning it has six protons in its nucleus and, in its neutral state, six electrons surrounding the nucleus. These electrons are distributed in specific energy levels or shells according to the principles of quantum mechanics. The electronic configuration of carbon is 1s²2s²2p².
- The 1s² shell: This innermost shell holds two electrons tightly bound to the nucleus. These electrons are generally considered too tightly bound to participate directly in chemical bonding. They are core electrons.
- The 2s² and 2p² shells: This second shell is where the action happens. It contains the valence electrons – those involved in forming chemical bonds. This shell has a total of four electrons: two in the 2s subshell and two in the 2p subshell.
Therefore, carbon has four valence electrons available for chemical bonding. This seemingly simple fact underpins carbon's extraordinary ability to form a vast array of molecules.
The Significance of Four Valence Electrons
The presence of four valence electrons dictates several crucial aspects of carbon's chemistry:
-
Tetravalency: Carbon's four valence electrons allow it to form four covalent bonds. A covalent bond is formed when two atoms share a pair of electrons. This tetravalency is the cornerstone of carbon's ability to form long chains, branched structures, and rings, making it ideal for creating the complex molecules that form the basis of life.
-
Versatile Bonding: Carbon can form single, double, and triple bonds with other carbon atoms and various other elements like hydrogen, oxygen, nitrogen, sulfur, and halogens. This ability to form multiple bond types contributes significantly to the diversity of organic molecules. Single bonds share one pair of electrons, double bonds share two pairs, and triple bonds share three pairs.
-
Catination: The remarkable capacity of carbon atoms to bond extensively with other carbon atoms is known as catenation. This characteristic is responsible for the existence of long chains, branched chains, and cyclic structures of carbon atoms, leading to the vastness of organic compounds.
Hybridisation: Expanding Carbon's Bonding Possibilities
While the simple electronic configuration suggests four separate orbitals, the reality is more nuanced. Carbon frequently undergoes hybridization, a process where atomic orbitals combine to form new hybrid orbitals with different shapes and energies. This hybridization significantly influences the geometry and properties of the molecules carbon forms.
-
sp³ Hybridization: In this type of hybridization, one 2s orbital and three 2p orbitals combine to form four equivalent sp³ hybrid orbitals. These orbitals are arranged tetrahedrally, with bond angles of approximately 109.5°. This hybridization is common in molecules like methane (CH₄) and ethane (C₂H₆).
-
sp² Hybridization: Here, one 2s orbital and two 2p orbitals combine to form three equivalent sp² hybrid orbitals, which are arranged in a trigonal planar geometry with bond angles of approximately 120°. The remaining unhybridized 2p orbital is involved in the formation of a π (pi) bond, resulting in a double bond. This hybridization is characteristic of molecules like ethene (C₂H₄).
-
sp Hybridization: In sp hybridization, one 2s orbital and one 2p orbital combine to form two equivalent sp hybrid orbitals, arranged linearly with a bond angle of 180°. The two remaining unhybridized 2p orbitals form two π bonds, resulting in a triple bond. This type of hybridization is observed in molecules like ethyne (C₂H₂).
Carbon's Role in Organic Chemistry and Beyond
The unique bonding capabilities of carbon, stemming directly from its four valence electrons, are the foundation of organic chemistry. It's the central element in:
-
Hydrocarbons: These are organic compounds consisting solely of carbon and hydrogen atoms. They form the basis for many fuels and are vital building blocks for more complex organic molecules.
-
Carbohydrates: These essential biological molecules provide energy and structural support in living organisms. They are composed of carbon, hydrogen, and oxygen.
-
Proteins: Proteins are complex molecules crucial for numerous biological functions. They consist of amino acids linked together, each amino acid containing a carbon backbone.
-
Lipids: Lipids include fats, oils, and waxes. They play vital roles in energy storage, cell membranes, and hormone production. Their structure is based on long hydrocarbon chains with oxygen and other elements incorporated.
-
Nucleic Acids: DNA and RNA, the carriers of genetic information, are complex polymers built from nucleotides, each nucleotide containing a carbon-based sugar.
Beyond the realm of biology, carbon's versatility extends to materials science, where it forms the basis of:
-
Diamonds: The strongest naturally occurring material, diamonds consist of a network of carbon atoms bonded tetrahedrally in a strong three-dimensional lattice.
-
Graphite: Graphite is a layered material with carbon atoms arranged in planar sheets. Its ability to conduct electricity makes it suitable for various applications.
-
Fullerenes: These molecules consist of carbon atoms arranged in spherical or tubular structures, exhibiting unique electronic and structural properties with potential applications in nanotechnology.
-
Graphene: A single layer of graphite, graphene is a remarkable material with exceptional electrical conductivity, strength, and flexibility, sparking immense interest for various technological applications.
Conclusion: The Ubiquity of Carbon
The seemingly simple fact that carbon has four valence electrons available for chemical bonding is responsible for its extraordinary versatility and its central role in the chemistry of life and materials. Its ability to form diverse molecules through single, double, and triple bonds, along with its capacity for catenation and hybridization, makes carbon the unique and essential element that it is. The exploration and understanding of carbon's bonding capabilities remain at the forefront of scientific advancements, constantly revealing new possibilities in diverse fields. The future of materials science, nanotechnology, and even our understanding of life itself are intrinsically linked to the fascinating properties of this remarkable element.
Latest Posts
Latest Posts
-
What Is The Fraction For 5
Apr 05, 2025
-
32 Is What Percent Of 50
Apr 05, 2025
-
Anti Derivative Of Square Root Of X
Apr 05, 2025
-
Greatest Common Factor Of 24 And 36
Apr 05, 2025
-
What Is 3 4 5 As A Decimal
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about How Many Electrons Does Carbon Have Available For Chemical Bonding . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
