Is Air A Compound Or Element Or Mixture
listenit
Apr 04, 2025 · 6 min read
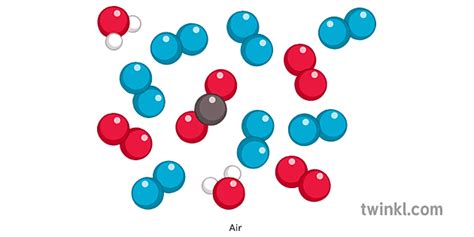
Table of Contents
Is Air a Compound, an Element, or a Mixture?
The question of whether air is a compound, an element, or a mixture is a fundamental one in chemistry, and understanding the answer requires a clear grasp of these three classifications of matter. While it might seem like a simple question, exploring the answer unveils a fascinating journey into the composition of our atmosphere and the nature of matter itself. This article will delve deep into the scientific definition of each term – element, compound, and mixture – before definitively answering the question regarding air and exploring its diverse components in detail. We will also touch upon the implications of air's classification on various aspects of our lives.
Understanding the Basic Classifications of Matter
Before we classify air, let's establish a solid understanding of the three categories: elements, compounds, and mixtures.
Elements: The Building Blocks of Matter
Elements are pure substances that cannot be broken down into simpler substances by chemical means. They are the fundamental building blocks of all matter. Each element is characterized by its unique atomic number, which represents the number of protons in the nucleus of its atoms. The periodic table organizes these elements, showcasing their properties and relationships. Examples of elements include oxygen (O), hydrogen (H), nitrogen (N), and iron (Fe). These elements are composed of only one type of atom.
Compounds: Elements Combined
Compounds are pure substances formed when two or more elements chemically combine in fixed proportions. This chemical combination results in a new substance with properties distinctly different from the elements that constitute it. The elements in a compound are held together by chemical bonds, such as ionic or covalent bonds. A crucial aspect is that the ratio of elements in a compound is always constant. For example, water (H₂O) is a compound composed of two hydrogen atoms and one oxygen atom in a fixed 2:1 ratio. You cannot change this ratio and still have water. Breaking down a compound requires a chemical reaction, not a simple physical separation.
Mixtures: A Blend of Substances
Mixtures, unlike elements and compounds, are not chemically bonded. They are physical combinations of two or more substances, where each substance retains its individual chemical properties. The components of a mixture can be separated by physical methods, such as filtration, distillation, or evaporation. Mixtures can have variable compositions; the ratio of components is not fixed. Examples include saltwater (a mixture of salt and water), air (a mixture of gases), and soil (a mixture of minerals and organic matter).
Air: A Definitive Classification
Now that we have a firm grasp of the distinctions between elements, compounds, and mixtures, we can definitively classify air. Air is a mixture. It's a homogeneous mixture, meaning its components are evenly distributed throughout, unlike a heterogeneous mixture such as sand and water, where the components are visibly distinct.
The Components of Air: A Deeper Dive
Air is primarily composed of several gases, each contributing to its properties and playing a critical role in various Earthly processes. Let's examine the major components:
Nitrogen (N₂): The Most Abundant Component
Nitrogen constitutes approximately 78% of Earth's atmosphere by volume. It's a relatively inert gas, meaning it doesn't readily react with other substances. While essential for life, organisms cannot directly utilize atmospheric nitrogen. Instead, it must be converted into usable forms through biological processes like nitrogen fixation.
Oxygen (O₂): Essential for Life
Oxygen makes up about 21% of the atmosphere and is vital for respiration in most living organisms. It's highly reactive, participating in numerous combustion and oxidation processes. The oxygen we breathe is crucial for cellular respiration, the process that releases energy from food.
Argon (Ar): An Inert Noble Gas
Argon is the third most abundant gas in the atmosphere, accounting for about 0.93%. It's a noble gas, meaning it's very unreactive and doesn't typically form compounds. Its inert nature makes it useful in various industrial applications.
Other Gases: Trace Amounts with Significant Impacts
Besides nitrogen, oxygen, and argon, air contains trace amounts of other gases, including:
- Carbon Dioxide (CO₂): Though present in relatively small amounts (around 0.04%), carbon dioxide plays a critical role in the Earth's climate through the greenhouse effect. Increased levels of CO₂ contribute to global warming.
- Neon (Ne), Helium (He), Methane (CH₄), Krypton (Kr), Hydrogen (H₂), and Xenon (Xe): These gases are present in even smaller concentrations but still contribute to the overall composition of the atmosphere. Some, like methane, are potent greenhouse gases.
- Water Vapor (H₂O): The amount of water vapor in the air is highly variable, depending on factors like temperature and location. It plays a crucial role in weather patterns and the water cycle.
Variable Composition: A Key Characteristic of Mixtures
The proportions of these gases in air can vary slightly depending on location, altitude, and other environmental factors. This variability is a hallmark of mixtures, further solidifying the classification of air as a mixture and not a compound. A compound always has a fixed ratio of its constituent elements.
Implications of Air's Classification
Understanding that air is a mixture has several important implications:
- Separation of Components: The fact that air is a mixture allows for the separation of its components through physical processes. For example, fractional distillation is used to separate the components of liquefied air, producing pure nitrogen, oxygen, and other gases used in various industrial applications.
- Pollution and Environmental Concerns: Because air is a mixture, pollutants can be easily incorporated into it. This highlights the importance of monitoring air quality and implementing measures to reduce pollution. The impact of greenhouse gases, which are components of the air mixture, is a pressing global concern.
- Respiratory Physiology: The composition of air directly affects respiratory function. The precise balance of oxygen and other gases is essential for proper breathing and overall health.
- Atmospheric Science: The study of the atmosphere, including its composition and dynamics, is a critical area of scientific research. Understanding the interactions between the different components of air is essential for comprehending climate change, weather patterns, and other atmospheric phenomena.
Conclusion: Air as a Dynamic Mixture
In conclusion, air is unequivocally a mixture, not a compound or an element. It's a dynamic and complex mixture of gases, each playing a vital role in maintaining the Earth's environment and supporting life. The variable composition and the ability to separate its components through physical means further confirm its classification as a mixture. This understanding is crucial for addressing environmental challenges, understanding atmospheric processes, and advancing scientific knowledge. The study of air, its composition, and its impact on our planet, will continue to be a critical area of focus in the years to come. The more we understand the complexities of this seemingly simple mixture, the better equipped we are to address the challenges and opportunities that come with it.
Latest Posts
Latest Posts
-
How Many Electrons Does Carbon Have Available For Chemical Bonding
Apr 04, 2025
-
What Is It Called When Everyone Plays Together
Apr 04, 2025
-
Y Is Inversely Proportional To X
Apr 04, 2025
-
Is Baking Bread A Chemical Or Physical Change
Apr 04, 2025
-
What Is The Half Life Of Potassium 40
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Is Air A Compound Or Element Or Mixture . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
