What Is The Half Life Of Potassium 40
listenit
Apr 04, 2025 · 6 min read
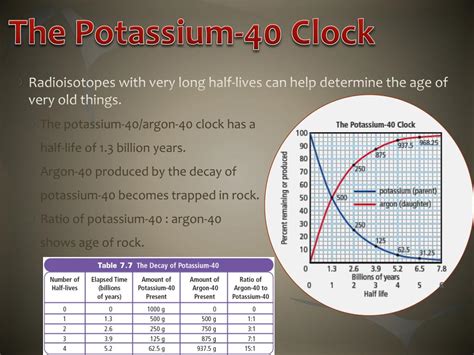
Table of Contents
What is the Half-Life of Potassium-40? A Deep Dive into Radioactive Decay
Potassium-40 (⁴⁰K) is a naturally occurring radioisotope of potassium, meaning it's a radioactive form of the element. Understanding its half-life is crucial in various scientific fields, from geological dating to medical imaging and even assessing radiation exposure. This article provides a comprehensive overview of potassium-40's half-life, its decay mechanisms, and its significance in different contexts.
Understanding Half-Life
Before delving into the specifics of potassium-40, let's define half-life. Half-life is the time it takes for half of the atoms in a radioactive sample to decay into a more stable form. It's a fundamental concept in nuclear physics and is a constant for a given radioisotope; it doesn't depend on external factors like temperature or pressure. Each radioactive element has its own unique half-life, ranging from fractions of a second to billions of years.
The Half-Life of Potassium-40: A Significant Timescale
The half-life of potassium-40 is approximately 1.251 × 10⁹ years, or 1.251 billion years. This incredibly long half-life is significant for several reasons:
-
Geological Dating: The long half-life makes ⁴⁰K ideal for radiometric dating of rocks and minerals, especially those older than a few million years. Techniques like potassium-argon dating rely on the decay of ⁴⁰K to its stable daughter isotope, ⁴⁰Ar (argon-40). By measuring the ratio of ⁴⁰K to ⁴⁰Ar in a sample, scientists can estimate the age of the material. This is invaluable for understanding Earth's geological history and the ages of ancient artifacts.
-
Abundance in the Earth's Crust: Potassium is a relatively abundant element in the Earth's crust, and a small fraction of that potassium is ⁴⁰K. Although the decay rate is slow, the sheer quantity of ⁴⁰K in the Earth contributes significantly to the planet's overall radioactivity, which plays a role in various geological processes.
-
Biological Significance: Potassium is essential for life, playing a critical role in cellular processes. Because ⁴⁰K is present in all living organisms, it contributes to a small but measurable amount of internal radiation exposure. While this radiation is generally considered harmless at low levels, understanding the decay rate is crucial in assessing potential risks in higher-exposure scenarios.
Potassium-40 Decay Modes: Beta Decay and Electron Capture
Unlike some radioisotopes that decay through a single pathway, ⁴⁰K undergoes two different decay modes:
1. Beta Decay (β- decay):
In beta decay, a neutron in the ⁴⁰K nucleus transforms into a proton, emitting a beta particle (an electron) and an antineutrino. This process results in the formation of calcium-40 (⁴⁰Ca), a stable isotope. Approximately 89.28% of ⁴⁰K decays through this method.
⁴⁰K → ⁴⁰Ca + β⁻ + ν̅ₑ
Where:
- ⁴⁰K is potassium-40
- ⁴⁰Ca is calcium-40
- β⁻ is a beta particle (electron)
- ν̅ₑ is an electron antineutrino
2. Electron Capture (EC):
Electron capture is a less common decay mode for ⁴⁰K, accounting for about 10.72% of its decays. In this process, the ⁴⁰K nucleus captures an inner-shell electron, converting a proton into a neutron. This results in the formation of argon-40 (⁴⁰Ar), another stable isotope. The captured electron combines with a proton to form a neutron and emits a neutrino.
⁴⁰K + e⁻ → ⁴⁰Ar + νₑ
Where:
- ⁴⁰K is potassium-40
- e⁻ is an electron
- ⁴⁰Ar is argon-40
- νₑ is an electron neutrino
The branching ratios (percentage of decays via each mode) are essential in determining the accurate age of geological samples using potassium-argon dating. The different decay products contribute to the overall potassium-40 decay scheme.
Implications of Potassium-40's Half-Life Across Disciplines
The long half-life of ⁴⁰K has profound implications across diverse scientific fields:
1. Geology and Geochronology:
As mentioned earlier, potassium-argon dating, using the ratio of ⁴⁰K to ⁴⁰Ar, is a cornerstone technique in geochronology. This method has been instrumental in establishing the ages of rocks and minerals, providing crucial insights into the Earth's formation and evolution. Other methods like potassium-calcium dating also utilize the decay of ⁴⁰K. The accuracy of these dating methods is directly linked to the precisely known half-life of ⁴⁰K.
2. Cosmochemistry and Planetary Science:
⁴⁰K's decay is also relevant in studying the composition and formation of planets and meteorites. Measuring the isotopic ratios of potassium and its decay products can help understand the processes that shaped these celestial bodies and constrain the age of the solar system. The heat generated by the decay of ⁴⁰K within planets contributes to their internal temperatures and geological activity.
3. Biology and Medicine:
Although the radiation from ⁴⁰K in biological systems is low, its presence is still significant. The internal radiation dose from ⁴⁰K is a factor considered in radiation protection and risk assessment. Medical imaging techniques, while not directly utilizing ⁴⁰K's decay, often rely on similar principles of radioactive decay and half-life for accurate diagnostic measurements. Understanding the radiation emitted by ⁴⁰K is crucial in medical and environmental monitoring.
4. Nuclear Physics and Radiation Safety:
The study of ⁴⁰K's decay contributes to a broader understanding of nuclear physics, radioactive decay mechanisms, and radiation safety. Accurate measurement of its half-life refines models of nuclear processes and helps in developing radiation safety protocols for handling radioactive materials. The research into ⁴⁰K contributes to the overall knowledge base for dealing with radioactive substances safely and responsibly.
Refining the Half-Life Measurement: Ongoing Research
The half-life of ⁴⁰K is not a fixed, immutable number. Scientists continually refine the measurement using advanced techniques to improve accuracy. Slight variations in reported values may arise from different measurement methods and the inherent uncertainties associated with such complex processes. However, the consensus value of 1.251 × 10⁹ years is widely accepted and used in scientific calculations. Ongoing research aims to further reduce the uncertainty in this crucial constant. Improved techniques and data analysis lead to more precise values, thus improving the reliability of dating methods and other applications dependent on the half-life of ⁴⁰K.
Conclusion
The half-life of potassium-40 (1.251 × 10⁹ years) is a fundamental constant with far-reaching implications in various scientific disciplines. Its long decay time makes it a valuable tool for geological dating, while its decay mechanisms provide insights into nuclear physics. The presence of ⁴⁰K in biological systems also necessitates consideration in radiation safety and risk assessments. As scientists continue to refine the measurement of its half-life and explore its multifaceted roles, our understanding of Earth's history, planetary science, and radiation effects will continue to improve. The ongoing study of ⁴⁰K underscores its importance as a key radioisotope in many aspects of science and technology.
Latest Posts
Latest Posts
-
What Is 0 3 Repeating As A Fraction
Apr 05, 2025
-
Common Factors Of 4 And 10
Apr 05, 2025
-
What Is Five Divided By Zero
Apr 05, 2025
-
Convert 75 Degrees Fahrenheit To Celsius
Apr 05, 2025
-
3 Gallons Equals How Many Pints
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about What Is The Half Life Of Potassium 40 . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
