The Monomers Of Proteins Are Called
listenit
Apr 02, 2025 · 6 min read
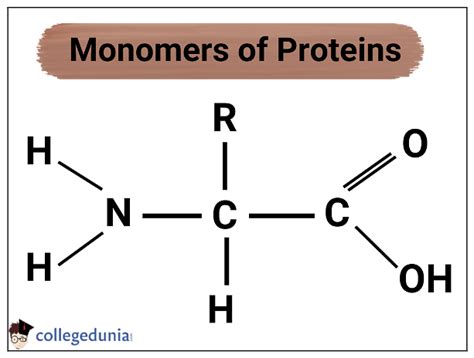
Table of Contents
The Monomers of Proteins Are Called: Amino Acids – A Deep Dive into Protein Structure and Function
Proteins are the workhorses of the cell, carrying out a vast array of functions crucial for life. From catalyzing biochemical reactions as enzymes to providing structural support and transporting molecules, proteins are indispensable. Understanding their structure is key to understanding their function, and this all begins with their fundamental building blocks: amino acids.
What are Amino Acids?
The monomers of proteins are indeed amino acids. These are organic molecules containing a central carbon atom (the α-carbon) bonded to four distinct chemical groups:
- An amino group (-NH₂): This group is basic and gives amino acids their name.
- A carboxyl group (-COOH): This group is acidic.
- A hydrogen atom (-H): A simple hydrogen atom.
- A side chain (R-group): This is the variable group that distinguishes one amino acid from another. The R-group's properties (size, charge, polarity, etc.) determine the amino acid's characteristics and ultimately influence the protein's overall structure and function.
The Diverse World of Amino Acid Side Chains
The 20 standard amino acids found in proteins differ solely in their R-groups. These variations lead to a remarkable diversity in protein structure and function. We can categorize amino acids based on their R-group properties:
1. Nonpolar, Aliphatic Amino Acids:
These amino acids have hydrophobic (water-fearing) side chains, often composed of hydrocarbon groups. Examples include:
- Glycine (Gly, G): The simplest amino acid, with a hydrogen atom as its R-group. Its small size allows for greater flexibility in protein folding.
- Alanine (Ala, A): Has a methyl group as its R-group.
- Valine (Val, V), Leucine (Leu, L), Isoleucine (Ile, I): These branched-chain amino acids are essential in the human diet, meaning our bodies cannot synthesize them.
2. Aromatic Amino Acids:
These amino acids possess aromatic rings in their side chains, contributing to their hydrophobic nature and ability to absorb ultraviolet light. Examples include:
- Phenylalanine (Phe, F): Contains a benzene ring.
- Tyrosine (Tyr, Y): Similar to phenylalanine, but with a hydroxyl group (-OH) on the benzene ring, making it slightly polar.
- Tryptophan (Trp, W): Contains an indole ring.
3. Polar, Uncharged Amino Acids:
These amino acids have polar, but uncharged, side chains, making them hydrophilic (water-loving). They often participate in hydrogen bonding within proteins. Examples include:
- Serine (Ser, S), Threonine (Thr, T): Contain hydroxyl groups (-OH).
- Cysteine (Cys, C): Contains a sulfhydryl group (-SH), capable of forming disulfide bonds which are crucial for protein stability.
- Asparagine (Asn, N), Glutamine (Gln, Q): Contain amide groups.
4. Positively Charged (Basic) Amino Acids:
These amino acids have positively charged side chains at physiological pH. They are hydrophilic and often participate in ionic interactions within proteins. Examples include:
- Lysine (Lys, K): Contains an amino group at the end of its side chain.
- Arginine (Arg, R): Contains a guanidinium group.
- Histidine (His, H): Contains an imidazole ring, which can act as both an acid and a base.
5. Negatively Charged (Acidic) Amino Acids:
These amino acids have negatively charged side chains at physiological pH. They are hydrophilic and participate in ionic interactions. Examples include:
- Aspartic acid (Asp, D): Contains a carboxyl group.
- Glutamic acid (Glu, E): Contains a carboxyl group.
Peptide Bonds: Linking Amino Acids Together
Amino acids are linked together by peptide bonds, a type of covalent bond. This process is called peptide synthesis and occurs through a dehydration reaction where a water molecule is removed. The carboxyl group of one amino acid reacts with the amino group of another, forming a peptide bond (-CO-NH-) and releasing a water molecule.
A chain of amino acids linked by peptide bonds is called a polypeptide. Proteins are essentially one or more polypeptide chains folded into a specific three-dimensional structure.
Levels of Protein Structure: From Primary to Quaternary
The three-dimensional structure of a protein is crucial for its function. This structure is hierarchical and can be described at four levels:
1. Primary Structure:
The primary structure is simply the linear sequence of amino acids in a polypeptide chain. This sequence is dictated by the genetic code, and any change in this sequence (e.g., a single amino acid substitution) can significantly alter the protein's structure and function. This is evident in genetic diseases like sickle cell anemia, where a single amino acid change in hemoglobin leads to a dysfunctional protein.
2. Secondary Structure:
Secondary structure refers to local folding patterns within the polypeptide chain. These are stabilized by hydrogen bonds between the carbonyl oxygen of one amino acid and the amide hydrogen of another. Common secondary structures include:
- α-helices: A right-handed coil structure.
- β-sheets: Extended strands arranged side-by-side, stabilized by hydrogen bonds between adjacent strands. These can be parallel or antiparallel depending on the orientation of the strands.
- Turns and loops: These are short regions connecting α-helices and β-sheets.
3. Tertiary Structure:
Tertiary structure refers to the overall three-dimensional arrangement of a single polypeptide chain. This structure is determined by interactions between the amino acid side chains, including:
- Hydrophobic interactions: Nonpolar side chains cluster together in the protein's interior, away from water.
- Hydrogen bonds: Form between polar side chains.
- Ionic bonds (salt bridges): Form between oppositely charged side chains.
- Disulfide bonds: Covalent bonds formed between cysteine residues.
The tertiary structure determines the protein's biological activity.
4. Quaternary Structure:
Quaternary structure refers to the arrangement of multiple polypeptide chains (subunits) in a protein complex. Not all proteins have quaternary structure; some are composed of only a single polypeptide chain. Interactions similar to those stabilizing tertiary structure also stabilize quaternary structure. A classic example is hemoglobin, which consists of four polypeptide subunits.
The Importance of Amino Acid Sequence in Protein Folding and Function
The precise sequence of amino acids dictates how a protein will fold. The sequence determines the distribution of hydrophobic and hydrophilic residues, which drives the folding process. Moreover, the specific arrangement of charged and polar amino acids determines the protein's interactions with other molecules and its overall function.
Changes in the amino acid sequence, even a single substitution, can drastically affect protein structure and function. This can lead to the formation of misfolded proteins, which can be associated with various diseases. For example, misfolded proteins are implicated in neurodegenerative diseases like Alzheimer's and Parkinson's.
Amino Acids and Human Health: Essential and Nonessential Amino Acids
The 20 standard amino acids can be classified as either essential or nonessential.
-
Essential amino acids: These cannot be synthesized by the human body and must be obtained from the diet. They include phenylalanine, valine, threonine, tryptophan, isoleucine, methionine, leucine, lysine, and histidine.
-
Nonessential amino acids: These can be synthesized by the human body from other metabolites. However, it is important to note that while the body can synthesize these amino acids, sufficient intake of precursors is still necessary for optimal synthesis.
A balanced diet containing sufficient amounts of all essential amino acids is crucial for proper protein synthesis and overall health.
Conclusion: Amino Acids – The Cornerstone of Life
Amino acids, the monomers of proteins, are fundamental to life. Their diverse chemical properties and the way they interact with each other lead to the vast array of protein structures and functions essential for all living organisms. Understanding the structure and properties of amino acids is key to comprehending the complex world of proteins and their crucial roles in biological processes. Further research into amino acids and protein folding continues to provide valuable insights into disease mechanisms and potential therapeutic strategies. The continued study of these fundamental building blocks of life remains a critical area of biological and medical research.
Latest Posts
Latest Posts
-
How Can A Sedimentary Rock Become A Metamorphic Rock
Apr 03, 2025
-
56 Is What Percent Of 80
Apr 03, 2025
-
The Triangles Are Similar What Is The Value Of X
Apr 03, 2025
-
A Baseball Leaves A Pitchers Hand Horizontally
Apr 03, 2025
-
What Is Ar On The Periodic Table
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about The Monomers Of Proteins Are Called . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
