What Is Ar On The Periodic Table
listenit
Apr 03, 2025 · 7 min read
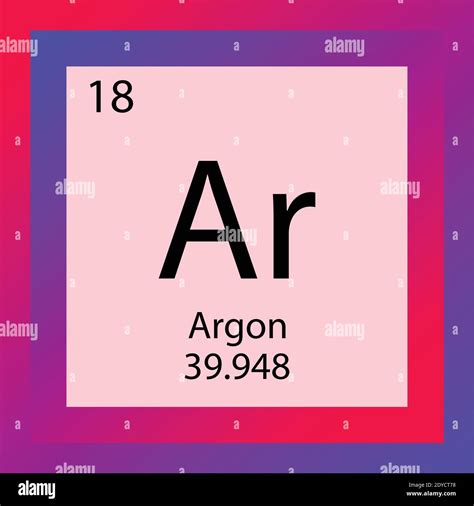
Table of Contents
What is Ar on the Periodic Table? Unveiling the Noble Gas Argon
Argon (Ar), atomic number 18, holds a significant place on the periodic table as a noble gas. This article delves deep into the properties, uses, discovery, and significance of argon, providing a comprehensive understanding of this often-overlooked element.
Understanding Argon's Position on the Periodic Table
Argon resides in Group 18, also known as the noble gases or inert gases. This group is characterized by elements with fully filled valence electron shells. This complete electron configuration contributes to their exceptional stability and low reactivity, a defining characteristic of noble gases. Argon, specifically, is located in Period 3, meaning it has three electron shells. This positioning dictates its atomic radius and other physical properties.
Key Features of Argon's Periodic Table Placement:
- Group 18 (Noble Gases): This placement immediately highlights argon's inert nature and its reluctance to participate in chemical reactions. Unlike elements in other groups that readily form bonds to achieve a stable electron configuration, argon already possesses this stability.
- Period 3: This indicates that argon has three electron shells. This influences its atomic size and the energy levels of its electrons. Elements in the same period share the same number of electron shells but have varying numbers of protons and electrons, leading to differences in their chemical and physical properties.
- Atomic Number 18: This signifies that a neutral argon atom contains 18 protons and 18 electrons. The number of protons defines the element and its position on the periodic table.
Argon's Physical and Chemical Properties
Argon's unique properties stem directly from its electron configuration and position on the periodic table. Understanding these properties is crucial to comprehending its various applications.
Physical Properties:
- Gas at Room Temperature: Argon exists as a colorless, odorless, and tasteless gas under standard conditions. Its gaseous state is a direct consequence of the weak interatomic forces between argon atoms.
- Low Density: Argon is denser than air, a property utilized in some of its applications. This higher density compared to other atmospheric gases is due to its atomic mass.
- Low Boiling and Melting Points: Argon has extremely low boiling and melting points, reflecting the weak intermolecular forces between its atoms. This makes it relatively easy to liquefy and solidify under appropriate conditions.
- Non-Conductive: Argon, like other noble gases, is an excellent electrical insulator, making it suitable for applications requiring electrical insulation.
- Monoatomic: Argon exists as a monoatomic gas, meaning it exists as individual atoms rather than forming molecules. This further emphasizes its inert nature.
Chemical Properties:
- Inertness: Argon's defining chemical characteristic is its inertness. Its fully filled valence shell makes it exceptionally stable and unreactive. It rarely forms chemical compounds. While some compounds have been synthesized under extreme conditions, these are rare and unstable.
- Low Reactivity: The reluctance of argon to participate in chemical reactions stems from its stable electron configuration. It has no tendency to gain, lose, or share electrons to achieve a more stable state.
- Ionization Energy: Argon possesses a relatively high ionization energy, meaning it requires a significant amount of energy to remove an electron from its outer shell. This further supports its inertness.
The Discovery of Argon: A Milestone in Chemistry
The discovery of argon stands as a testament to scientific curiosity and meticulous experimentation. Unlike many elements discovered through straightforward chemical reactions, argon's discovery relied on careful observation and innovative techniques.
Lord Rayleigh and Sir William Ramsay: The Pioneers
Lord Rayleigh, a British physicist, noticed inconsistencies in the density of nitrogen gas prepared from different sources. Nitrogen obtained from the air was consistently denser than nitrogen prepared from chemical compounds. This discrepancy intrigued him, leading him to collaborate with Sir William Ramsay, a Scottish chemist.
Fractional Distillation: The Key Technique
Their collaborative research utilized fractional distillation of liquefied air. This process separates components of a mixture based on their boiling points. By carefully fractionating liquefied air, they isolated a residual gas that was denser than nitrogen and chemically inert. This inert gas was identified as a new element – argon.
Significance of Argon's Discovery:
Argon's discovery was pivotal for several reasons:
- Challenged Existing Chemical Theories: The discovery of an inert gas contradicted the existing belief that all elements could form chemical compounds. It paved the way for a deeper understanding of chemical bonding and the periodic table.
- Expanded the Periodic Table: Argon's discovery expanded the periodic table and led to the recognition of the noble gas group.
- Opened New Research Avenues: The isolation of argon spurred further research into noble gases, ultimately leading to the discovery of other noble gases like helium, neon, krypton, xenon, and radon.
Applications of Argon: Diverse Uses Across Industries
Argon's unique properties make it a versatile element with applications across numerous industries. Its inertness and other characteristics contribute to its widespread use.
Industrial Applications:
- Welding and Metal Fabrication: Argon's inertness protects the weld from oxidation and contamination, leading to stronger and more durable welds. It's used as a shielding gas in various welding processes, including Gas Metal Arc Welding (GMAW) and Gas Tungsten Arc Welding (GTAW).
- Metal Production: Argon is employed in the production of reactive metals like titanium and zirconium. Its inert atmosphere prevents oxidation during the refining process.
- Chemical Processing: Argon serves as an inert atmosphere in chemical reactors, preventing unwanted reactions. This is particularly important when handling reactive chemicals.
- Electronics Manufacturing: Argon is used in the manufacturing of semiconductor devices and other electronic components. It provides a clean and inert environment for fabrication processes.
Other Applications:
- Light Bulbs: Argon is used as a filling gas in incandescent light bulbs. Its inertness prevents oxidation of the filament, extending its lifespan.
- Scientific Instruments: Argon's inertness makes it suitable for use in various scientific instruments, particularly those involving sensitive chemical reactions.
- Medical Applications: While less common than other noble gases, argon has some medical applications, such as in laser surgery.
- Food Packaging: Argon's inert nature helps maintain the quality and freshness of packaged foods. It displaces oxygen, preventing spoilage.
Environmental Considerations: Argon and Sustainability
Argon is widely regarded as an environmentally friendly element. Unlike some elements, argon does not pose significant environmental hazards.
Abundance and Extraction:
Argon is relatively abundant in the atmosphere, representing approximately 0.93% of the Earth's atmosphere. Its extraction is primarily achieved through the fractional distillation of liquefied air. This process is energy-intensive, but the overall environmental impact of argon production is relatively low.
Non-Toxicity and Biodegradability:
Argon is non-toxic and does not accumulate in the environment. It is inert and does not participate in chemical reactions that could harm ecosystems. Its inert nature renders it essentially harmless to living organisms.
The Future of Argon: Continued Importance and Potential Advancements
Argon’s importance is likely to remain constant or even increase in the future. Its inert nature and unique physical properties make it indispensable in various industrial processes and technological advancements.
Emerging Applications:
Potential areas where argon's use might expand include:
- Advanced Materials Science: Argon could play an increasing role in the development and processing of advanced materials with unique properties.
- Renewable Energy Technologies: Its inert properties could be beneficial in certain renewable energy technologies.
- Space Exploration: Argon's characteristics might find applications in space exploration and related technologies.
Conclusion: Argon – A Noble Gas with Significant Impact
Argon, with its unique position on the periodic table and resulting properties, stands as a fascinating element with widespread applications. Its inertness, coupled with its abundance and ease of extraction, makes it a valuable resource across diverse industries. From welding to electronics manufacturing and beyond, argon continues to play a vital role in modern technology and societal advancement. Its discovery significantly broadened our understanding of chemistry and continues to inspire ongoing research and development. As technology advances, the importance of argon and its various applications is likely to only increase.
Latest Posts
Latest Posts
-
Abiotic Features Of A Coral Reef
Apr 04, 2025
-
How Do You Find The Molar Mass Of A Gas
Apr 04, 2025
-
What Are The Horizontal Rows Called On The Periodic Table
Apr 04, 2025
-
Is The Median Of A Trapezoid Parallel To Eahc Base
Apr 04, 2025
-
Are Days Of The Week Proper Nouns
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about What Is Ar On The Periodic Table . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
