What Are The Horizontal Rows Called On The Periodic Table
listenit
Apr 04, 2025 · 6 min read
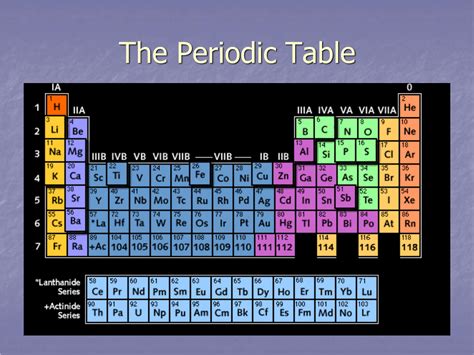
Table of Contents
What Are the Horizontal Rows Called on the Periodic Table?
The periodic table, a cornerstone of chemistry, organizes chemical elements in a structured format based on their atomic number, electron configuration, and recurring chemical properties. While the vertical columns are known as groups or families, the horizontal rows hold a different, equally important designation. This article delves deep into the nomenclature and significance of these horizontal rows, explaining what they are called, how they are organized, and the trends and patterns they reveal.
Understanding the Periodic Table's Structure: Rows vs. Columns
Before diving into the specifics of the horizontal rows, it's crucial to understand the overall structure of the periodic table. The table is arranged in a grid-like format, with elements organized both horizontally and vertically. These arrangements are not arbitrary but reflect fundamental properties of the elements.
-
Columns (Groups or Families): Elements within the same vertical column share similar chemical properties because they have the same number of valence electrons – the electrons in the outermost shell that participate in chemical bonding. These columns are numbered 1 through 18, and elements within each group exhibit predictable behaviors in chemical reactions.
-
Rows (Periods): Elements in the same horizontal row, the subject of this article, share the same highest principal quantum number (n). This principal quantum number corresponds to the energy level of the outermost electrons. As we move across a period, the number of protons and electrons increases, leading to a systematic change in the element's properties.
The Name: Periods
The answer to the question "What are the horizontal rows called on the periodic table?" is simple: they are called periods. Each period represents a principal energy level or shell that is progressively filled with electrons as we move across the row. The number of elements in each period varies, reflecting the complexity of electron shell filling.
Why "Periods"?
The term "period" aptly describes the recurring patterns of properties observed as we traverse the horizontal rows. The properties generally follow a predictable trend within each period, starting with highly reactive metals, moving towards less reactive elements, then onto nonmetals, and finally ending with noble gases, which are largely inert. This cyclical repetition of properties across the periods is a key feature of the periodic table's organization.
The Significance of Periods: Trends and Patterns
The organization of elements into periods highlights several crucial trends and patterns in their properties:
1. Atomic Radius:
-
Across a Period (Left to Right): Atomic radius generally decreases across a period. This is because the addition of protons increases the nuclear charge, pulling the electrons closer to the nucleus, resulting in a smaller atomic size.
-
Down a Period (Top to Bottom): Atomic radius generally increases down a period. This occurs because additional electron shells are added, increasing the distance between the nucleus and the outermost electrons.
2. Ionization Energy:
-
Across a Period (Left to Right): Ionization energy generally increases across a period. It becomes increasingly difficult to remove an electron as the nuclear charge increases, holding the electrons more tightly.
-
Down a Period (Top to Bottom): Ionization energy generally decreases down a period. The increased distance between the nucleus and the outermost electrons makes it easier to remove an electron.
3. Electronegativity:
-
Across a Period (Left to Right): Electronegativity generally increases across a period. Elements on the right side of the periodic table have a stronger tendency to attract electrons in a chemical bond.
-
Down a Period (Top to Bottom): Electronegativity generally decreases down a period. The increased distance between the nucleus and the valence electrons reduces the attraction for bonding electrons.
4. Metallic Character:
-
Across a Period (Left to Right): Metallic character generally decreases across a period. Metals tend to be found on the left side, while nonmetals are on the right.
-
Down a Period (Top to Bottom): Metallic character generally increases down a period. Elements lower in the table exhibit more metallic characteristics.
The Number of Periods and their Elements
The periodic table currently comprises seven periods:
-
Period 1: Contains only two elements, hydrogen (H) and helium (He). This short period reflects the filling of the first electron shell (n=1), which can only hold a maximum of two electrons.
-
Period 2: Contains eight elements, from lithium (Li) to neon (Ne). This period corresponds to the filling of the second electron shell (n=2), which can hold up to eight electrons.
-
Period 3: Also contains eight elements, from sodium (Na) to argon (Ar). Similar to Period 2, it represents the filling of the third electron shell (n=3), which can also accommodate up to eight electrons.
-
Period 4: Contains 18 elements, from potassium (K) to krypton (Kr). This is where the d-block elements (transition metals) are introduced, reflecting the filling of the 3d subshell.
-
Period 5: Contains 18 elements, from rubidium (Rb) to xenon (Xe), mirroring the structure of Period 4, with the filling of the 4d subshell.
-
Period 6: Contains 32 elements, from cesium (Cs) to radon (Rn). This period includes the f-block elements (lanthanides and actinides), representing the filling of the 4f subshell. These elements are typically placed separately at the bottom of the periodic table to maintain a manageable table width.
-
Period 7: Currently incomplete, it is expected to contain 32 elements, mirroring period 6 in structure. The filling of the 5f subshell continues, and many of the elements in this period are synthetically produced radioactive elements.
Beyond the Basics: Understanding Subshells and Electron Configuration
The organization of elements into periods is directly linked to their electron configurations. Each period corresponds to the filling of a principal electron shell (n=1, 2, 3, etc.). However, the nuances become more complex when considering subshells (s, p, d, f).
-
s-block elements: The first two columns of the periodic table (Groups 1 and 2) represent the filling of the s subshell. These elements are generally highly reactive metals.
-
p-block elements: The last six columns of the periodic table (Groups 13-18) represent the filling of the p subshell. This block contains a mix of metals, metalloids, and nonmetals, showcasing a diverse range of properties.
-
d-block elements (transition metals): The ten columns in the middle of the periodic table (Groups 3-12) represent the filling of the d subshell. Transition metals exhibit variable oxidation states and form complex ions, contributing to their significant role in chemistry and industry.
-
f-block elements (inner transition metals): The lanthanides (rare earth elements) and actinides are placed separately at the bottom of the periodic table. They represent the filling of the f subshell, and many are radioactive.
Understanding these subshells and their filling order is crucial for comprehending the electron configurations of elements and their resulting properties within each period.
The Periodic Table: A Dynamic and Evolving Tool
The periodic table is not a static document. As our understanding of chemistry and the properties of elements deepens, the table continues to evolve. The discovery of new elements and the refinement of our theoretical understanding lead to minor adjustments and reinterpretations. The fundamental principle of organizing elements based on their atomic number and recurring chemical properties remains, however, the backbone of this invaluable tool.
Conclusion: The Importance of Understanding Periods
The horizontal rows, known as periods, are a fundamental aspect of the periodic table's structure and organization. They reveal significant trends and patterns in the properties of elements, reflecting the systematic filling of electron shells and subshells. By understanding the arrangement of elements within periods, chemists can predict and explain various chemical phenomena, and develop new materials and technologies based on the predictable properties of these elements. The periodic table, therefore, remains an indispensable tool for anyone seeking to explore the fascinating world of chemistry.
Latest Posts
Latest Posts
-
How Many Unpaired Electrons Does Phosphorus Have
Apr 04, 2025
-
How Many Protons Does Xenon Have
Apr 04, 2025
-
What Is The Decimal For 17 20
Apr 04, 2025
-
What Is The Name Of The Compound Ca No3 2
Apr 04, 2025
-
What Is The Percent Acetic Acid In Vinegar
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about What Are The Horizontal Rows Called On The Periodic Table . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
