The Change In State From Liquid To Gas Is Called
listenit
Apr 03, 2025 · 6 min read
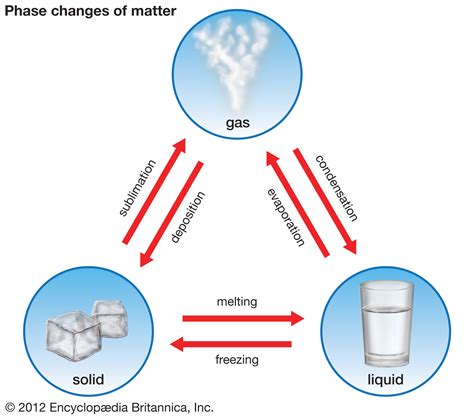
Table of Contents
The Change in State from Liquid to Gas is Called Vaporization: A Deep Dive
The transition of a substance from a liquid state to a gaseous state is a fundamental process in chemistry and physics, crucial to understanding weather patterns, industrial processes, and even biological functions. This process is known as vaporization. But vaporization is not a monolithic process; it encompasses several distinct mechanisms, each influenced by factors like temperature, pressure, and the specific properties of the substance. This article will explore the intricacies of vaporization, delving into its various forms, underlying principles, and practical applications.
Understanding the Phases of Matter and the Transition to Gas
Before we delve into the specifics of vaporization, it's important to establish a foundational understanding of the three primary states of matter: solid, liquid, and gas. These states are defined by the arrangement and energy levels of their constituent molecules.
-
Solids: In solids, molecules are tightly packed together in a fixed, ordered arrangement. They possess strong intermolecular forces, resulting in a rigid structure with a defined shape and volume.
-
Liquids: Liquids exhibit a less ordered structure than solids. Molecules are still relatively close together, but they can move and slide past each other, resulting in a defined volume but an indefinite shape. The intermolecular forces are weaker than in solids.
-
Gases: Gases are characterized by widely dispersed molecules with weak intermolecular forces. Molecules in a gaseous state are in constant, random motion, exhibiting neither a defined shape nor a defined volume. They readily fill the container they occupy.
The transition from a liquid to a gas involves overcoming the intermolecular forces holding the liquid molecules together, requiring an input of energy. This energy typically comes in the form of heat.
The Different Types of Vaporization
Vaporization isn't a single, uniform process. It's actually an umbrella term encompassing several distinct mechanisms:
1. Evaporation: A Gradual Process at the Surface
Evaporation is the gradual transformation of a liquid into a gas at the surface of the liquid. It occurs at temperatures below the boiling point. During evaporation, only the most energetic molecules, those with sufficient kinetic energy to overcome the intermolecular forces, escape from the liquid's surface and enter the gaseous phase. This process is significantly influenced by factors such as temperature, surface area, and air movement. Higher temperatures lead to faster evaporation, as more molecules possess the necessary kinetic energy. A larger surface area exposes more molecules to the surrounding environment, facilitating evaporation. Air movement (wind) also enhances evaporation by continuously removing the gaseous molecules from the surface, creating a concentration gradient that promotes further evaporation.
2. Boiling: A Violent Process Throughout the Liquid
Boiling, on the other hand, is a much more vigorous process. It occurs when the vapor pressure of the liquid equals the external pressure (typically atmospheric pressure). At this point, vapor bubbles begin to form within the liquid, not just at the surface. These bubbles rise to the surface and burst, releasing the gaseous phase into the atmosphere. The boiling point of a liquid is the temperature at which this occurs. Unlike evaporation, which is a surface phenomenon, boiling is a bulk phenomenon involving the entire volume of the liquid. The boiling point of a liquid depends on the external pressure; at higher pressures, the boiling point increases. This principle is exploited in pressure cookers, which allow for faster cooking by increasing the boiling point of water.
3. Sublimation (Solid to Gas): A Special Case
While not strictly a liquid-to-gas transition, it's important to mention sublimation, the process where a solid directly transforms into a gas without passing through the liquid phase. This occurs when the vapor pressure of the solid exceeds the external pressure, and the molecules have enough kinetic energy to escape directly from the solid lattice. Common examples include dry ice (solid carbon dioxide) and naphthalene (mothballs). While not directly related to the liquid-to-gas transition, it highlights the dynamic nature of phase changes.
Factors Affecting Vaporization
Several factors influence the rate of vaporization:
-
Temperature: Higher temperatures increase the kinetic energy of molecules, making it easier for them to overcome intermolecular forces and transition to the gaseous phase. This accelerates both evaporation and boiling.
-
Surface Area: A larger surface area provides more opportunities for molecules to escape the liquid phase, increasing the rate of evaporation.
-
Air Movement (Wind): Wind removes gaseous molecules from the vicinity of the liquid surface, reducing the concentration of the gas phase and accelerating evaporation.
-
Pressure: Lower external pressure reduces the boiling point, making it easier for the liquid to boil and vaporize. Higher external pressure increases the boiling point.
-
Intermolecular Forces: Stronger intermolecular forces require more energy to overcome, resulting in slower vaporization rates. Substances with weak intermolecular forces, such as volatile organic compounds, vaporize more readily than substances with strong intermolecular forces, such as water.
-
Humidity: High humidity (high concentration of water vapor in the air) slows down evaporation, as the air is already saturated with water vapor.
Practical Applications of Vaporization
Vaporization plays a crucial role in numerous applications across various fields:
-
Cooling Systems: Evaporation is used in cooling systems like refrigerators and air conditioners. The evaporation of a refrigerant absorbs heat from the surroundings, causing cooling.
-
Distillation: This process separates liquids based on their different boiling points. Vaporization and condensation are key steps in separating components of a mixture.
-
Power Generation: Steam turbines in power plants utilize the vaporization of water to generate electricity.
-
Weather Patterns: Evaporation from oceans, lakes, and rivers is a fundamental part of the water cycle and drives weather patterns.
-
Industrial Processes: Vaporization is used in numerous industrial processes, such as drying, solvent recovery, and chemical reactions.
-
Medical Applications: Vaporization is used in certain medical treatments, such as nebulizers for administering medication.
The Clausius-Clapeyron Equation: A Quantitative Approach
The Clausius-Clapeyron equation provides a quantitative relationship between the vapor pressure of a liquid and temperature:
ln(P2/P1) = -ΔHvap/R * (1/T2 - 1/T1)
Where:
- P1 and P2 are the vapor pressures at temperatures T1 and T2 respectively.
- ΔHvap is the enthalpy of vaporization (the energy required to vaporize one mole of liquid).
- R is the ideal gas constant.
This equation allows for the prediction of vapor pressure at different temperatures, a crucial parameter in understanding and controlling vaporization processes.
Conclusion: A Multifaceted Process with Wide-Ranging Implications
The change in state from liquid to gas, known as vaporization, is a complex yet fascinating process with profound implications across various fields. Understanding the different types of vaporization – evaporation and boiling – and the factors influencing these processes is crucial for applications ranging from everyday life to sophisticated industrial processes. The Clausius-Clapeyron equation provides a powerful tool for quantitatively analyzing vaporization phenomena. Further research into the intricacies of vaporization continues to unlock new possibilities and enhance our understanding of the world around us. From the subtle cooling effect of evaporation on a hot day to the powerful force of boiling water used to generate electricity, vaporization is a process that shapes our world in numerous ways. Its study reveals the fundamental principles governing the behavior of matter and underpins a vast array of technological advancements and natural phenomena. The continued exploration of this process promises further advancements in various fields, expanding our understanding and capabilities.
Latest Posts
Latest Posts
-
How Did He Know That The Nucleus Was Positively Charged
Apr 03, 2025
-
What Is 4 To The 5 Power
Apr 03, 2025
-
Integral 1 X 2 3 2
Apr 03, 2025
-
Y 3 X 5 X 2
Apr 03, 2025
-
What Is The Value Of 1
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about The Change In State From Liquid To Gas Is Called . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
