Number Of Valence Electrons In Ar
listenit
Apr 02, 2025 · 5 min read
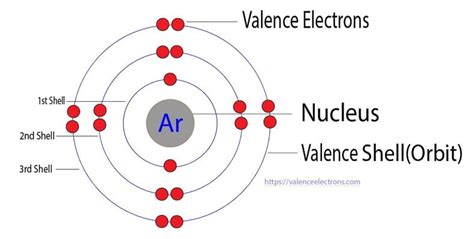
Table of Contents
Number of Valence Electrons in Argon: A Deep Dive into Atomic Structure and Chemical Properties
Argon (Ar), a noble gas residing in Group 18 of the periodic table, is renowned for its chemical inertness. This inertness stems directly from its electronic configuration, specifically the number of valence electrons it possesses. Understanding the number of valence electrons in Argon is crucial to grasping its unique chemical behavior and its applications in various fields. This article will delve deep into the atomic structure of Argon, explaining why it has the number of valence electrons it does and exploring the consequences of this configuration.
Understanding Valence Electrons
Before focusing on Argon, let's establish a fundamental understanding of valence electrons. Valence electrons are the outermost electrons in an atom's electron shell. These electrons are the ones involved in chemical bonding and determine an element's chemical reactivity. Atoms strive for stability, typically achieving it by having a full outermost shell – a configuration often referred to as a stable octet (eight electrons) or a duet (two electrons for elements like hydrogen and helium).
The number of valence electrons an atom possesses is directly related to its group number in the periodic table. For main group elements (Groups 1-18), the group number usually corresponds to the number of valence electrons. However, this rule has exceptions, particularly for transition metals.
Argon's Electronic Configuration and Valence Electrons
Argon's atomic number is 18, meaning it has 18 protons and 18 electrons in a neutral atom. These electrons are distributed across different energy levels or shells. The electronic configuration of Argon is 1s²2s²2p⁶3s²3p⁶. Let's break this down:
- 1s²: Two electrons in the first energy level (shell).
- 2s²: Two electrons in the second energy level.
- 2p⁶: Six electrons in the second energy level's p subshell.
- 3s²: Two electrons in the third energy level.
- 3p⁶: Six electrons in the third energy level's p subshell.
The outermost shell for Argon is the third energy level (n=3), containing the 3s and 3p electrons. Adding the electrons in these subshells (2 + 6 = 8), we find that Argon has 8 valence electrons.
The Significance of Eight Valence Electrons
The presence of eight valence electrons in Argon is the key to understanding its chemical inertness. This configuration represents a complete outermost shell, satisfying the octet rule. Atoms with a complete octet are exceptionally stable because they have minimal tendency to gain, lose, or share electrons to achieve a lower energy state. This stability translates into a very low reactivity.
Other noble gases, such as Helium (He), Neon (Ne), Krypton (Kr), Xenon (Xe), and Radon (Rn), also exhibit this characteristic of having a full outermost shell and consequently possess very low reactivity. This is why the noble gases are often called inert gases.
Exceptions to the Octet Rule: The Case of Helium
It's important to note that Helium, with only two electrons (1s²), is an exception to the octet rule. Helium's outermost shell is the first energy level, which can only hold a maximum of two electrons. With its full outermost shell, Helium, like other noble gases, is chemically inert. This highlights that a full outermost shell, regardless of whether it contains eight or two electrons, signifies stability and low reactivity.
Argon's Chemical Inertness and its Applications
Argon's chemical inertness is the foundation for its numerous applications in various industries:
1. Welding and Metallurgy:
Argon's inertness prevents the oxidation of molten metals during welding processes. It creates a protective atmosphere around the weld, preventing contamination and ensuring high-quality welds. This is crucial in industries such as automotive manufacturing, aerospace, and construction.
2. Lighting:
Argon is used in incandescent and fluorescent lighting. In incandescent bulbs, it slows down the evaporation of the tungsten filament, extending the bulb's lifespan. In fluorescent lamps, Argon's presence enhances the ionization process, making the lamp more efficient.
3. Electronics:
Argon is employed as a protective gas in the semiconductor industry, safeguarding sensitive electronic components from oxidation and contamination during manufacturing. This is vital for the production of microchips and other electronic devices.
4. Medical Applications:
Argon lasers are used in various medical procedures, including ophthalmology (eye surgery) and dermatology (skin treatments). The laser's precise action and Argon's inertness make it suitable for delicate surgical procedures.
5. Food Packaging:
In the food industry, Argon is used as a packaging gas to extend the shelf life of food products. Its inertness prevents the oxidation of food, maintaining its freshness and quality for longer periods.
Beyond the Basics: Exploring Argon's Isotopes and Nuclear Properties
While the chemical behavior of Argon is largely determined by its 8 valence electrons, its nuclear properties also contribute to its significance. Argon has several isotopes, meaning atoms with the same number of protons but a different number of neutrons. The most common isotope is Argon-40, accounting for over 99% of naturally occurring Argon. Understanding Argon's isotopes is crucial in various scientific fields, including geochronology and environmental science.
Argon-40 is a product of the radioactive decay of Potassium-40, a naturally occurring radioactive isotope. The ratio of Argon-40 to Potassium-40 in rocks is used to determine the age of geological formations, a technique known as Potassium-Argon dating. This dating method is a powerful tool in understanding Earth's history and the evolution of life.
Conclusion: Argon's Inertness, a Consequence of its Eight Valence Electrons
In summary, the number of valence electrons in Argon (8) is directly responsible for its chemical inertness and diverse applications. The complete octet in its outermost shell provides exceptional stability, limiting its tendency to participate in chemical reactions. This property makes Argon invaluable in various industrial and scientific processes, from welding to dating geological formations. Understanding Argon's atomic structure, including its electronic configuration and the significance of its valence electrons, allows us to appreciate the profound impact of this noble gas on diverse aspects of modern technology and scientific inquiry. The unique interplay between its chemical and nuclear properties continues to fuel research and innovation across numerous disciplines. Further research into Argon’s behaviour under extreme conditions, such as those found in astrophysical environments, continues to reveal new facets of this seemingly unremarkable yet scientifically significant element.
Latest Posts
Latest Posts
-
What Are The Monomers Of Protein Polymers
Apr 03, 2025
-
How To Find Relative Maxima And Minima
Apr 03, 2025
-
How Do I Convert 3 8 Into A Decimal
Apr 03, 2025
-
What Is The Value Of H 2
Apr 03, 2025
-
What Is The Common Factor Of 8 And 12
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Number Of Valence Electrons In Ar . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
