What Are The Monomers Of Protein Polymers
listenit
Apr 03, 2025 · 6 min read
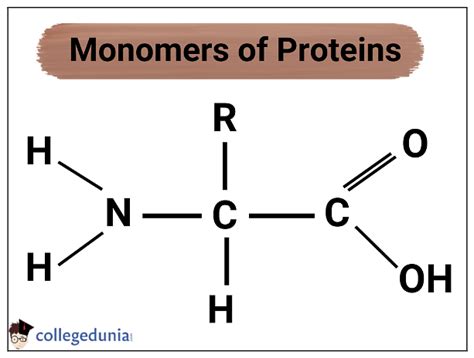
Table of Contents
What Are the Monomers of Protein Polymers?
Proteins are fundamental biomolecules essential for virtually every biological process. From catalyzing reactions to providing structural support, their diverse functions stem from their unique structures, which are ultimately dictated by their constituent monomers: amino acids. This article delves deep into the world of amino acids, exploring their structure, properties, and how they combine to form the incredible complexity of protein polymers. We'll also touch upon the implications of amino acid variations on protein structure and function.
Understanding Amino Acids: The Building Blocks of Proteins
Amino acids are organic molecules containing a central carbon atom (the α-carbon) bonded to four distinct groups:
- An amino group (-NH₂): This group is basic and carries a positive charge at physiological pH.
- A carboxyl group (-COOH): This group is acidic and carries a negative charge at physiological pH.
- A hydrogen atom (-H): A simple hydrogen atom.
- A side chain (R-group): This is the variable group that distinguishes one amino acid from another. The R-group's properties (size, charge, polarity, hydrophobicity) dictate the amino acid's characteristics and influence protein structure and function.
The general formula for an amino acid can be represented as: NH₂-CHR-COOH, where R represents the side chain.
The 20 Standard Amino Acids: A Diverse Group
There are 20 standard amino acids used by ribosomes to synthesize proteins. These amino acids are categorized based on their side chain properties:
1. Nonpolar, Aliphatic Amino Acids:
These amino acids have hydrocarbon side chains that are hydrophobic (water-repelling). Examples include:
- Glycine (Gly, G): The simplest amino acid, with a hydrogen atom as its side chain. Its small size allows for greater flexibility in protein structure.
- Alanine (Ala, A): A methyl group (-CH₃) as its side chain.
- Valine (Val, V): A branched isopropyl group (-CH(CH₃)₂) as its side chain.
- Leucine (Leu, L): A branched isobutyl group (-CH₂CH(CH₃)₂) as its side chain.
- Isoleucine (Ile, I): A branched sec-butyl group (-CH(CH₃)CH₂CH₃) as its side chain.
- Methionine (Met, M): Contains a thioether group (-CH₂CH₂SCH₃) in its side chain. It's often the initiator amino acid in protein synthesis.
2. Aromatic Amino Acids:
These amino acids possess aromatic rings in their side chains, making them relatively hydrophobic.
- Phenylalanine (Phe, F): Contains a benzene ring (-CH₂C₆H₅) in its side chain.
- Tyrosine (Tyr, Y): Similar to phenylalanine, but with a hydroxyl group (-OH) attached to the benzene ring, making it slightly more polar.
- Tryptophan (Trp, W): Contains an indole ring in its side chain. This large, bulky group significantly impacts protein folding.
3. Polar, Uncharged Amino Acids:
These amino acids have polar side chains that can participate in hydrogen bonding but do not carry a net charge at physiological pH.
- Serine (Ser, S): Contains a hydroxyl group (-OH) in its side chain.
- Threonine (Thr, T): Similar to serine, with a hydroxyl group on a β-carbon.
- Cysteine (Cys, C): Contains a thiol group (-SH) in its side chain. Two cysteine residues can form a disulfide bond, crucial for stabilizing protein structure.
- Asparagine (Asn, N): Contains an amide group (-CONH₂) in its side chain.
- Glutamine (Gln, Q): Contains a longer amide group (-CONH₂CH₂) in its side chain.
4. Positively Charged (Basic) Amino Acids:
These amino acids have positively charged side chains at physiological pH.
- Lysine (Lys, K): Contains an amino group (-NH₃⁺) at the end of its side chain.
- Arginine (Arg, R): Contains a guanidinium group (+H₂N=C(NH₂)NH-) in its side chain, which is highly basic.
- Histidine (His, H): Contains an imidazole ring in its side chain, which can act as both an acid and a base. Its pKa is close to physiological pH, making it important in enzyme catalysis.
5. Negatively Charged (Acidic) Amino Acids:
These amino acids have negatively charged side chains at physiological pH.
- Aspartic acid (Asp, D): Contains a carboxyl group (-COO⁻) in its side chain.
- Glutamic acid (Glu, E): Contains a longer carboxyl group (-COO⁻CH₂) in its side chain.
Peptide Bonds: Linking Amino Acids into Polymers
Amino acids are linked together via peptide bonds, which are amide bonds formed between the carboxyl group of one amino acid and the amino group of another. This reaction releases a molecule of water (dehydration synthesis). A chain of amino acids linked by peptide bonds is called a polypeptide.
The Primary Structure of Proteins: The Amino Acid Sequence
The linear sequence of amino acids in a polypeptide chain constitutes the primary structure of a protein. This sequence is determined by the genetic code and is crucial for determining the higher-order structures and ultimately the protein's function. Even a single amino acid change can drastically alter a protein's properties.
Higher-Order Structures: Secondary, Tertiary, and Quaternary Structures
The primary structure dictates how a polypeptide chain folds into its higher-order structures:
-
Secondary Structure: This refers to local folding patterns stabilized by hydrogen bonds between the backbone amide and carbonyl groups. Common secondary structures include α-helices and β-sheets.
-
Tertiary Structure: This describes the overall three-dimensional arrangement of a polypeptide chain, including interactions between side chains. These interactions include hydrophobic interactions, hydrogen bonds, ionic bonds, and disulfide bonds.
-
Quaternary Structure: This refers to the arrangement of multiple polypeptide chains (subunits) in a protein complex. Many proteins, such as hemoglobin, require multiple subunits to function correctly.
The Importance of Amino Acid Variation
The 20 standard amino acids, with their diverse side chain properties, provide the building blocks for an astonishing array of protein structures and functions. Variations in amino acid sequence lead to differences in:
-
Protein Folding: The hydrophobic and hydrophilic properties of side chains dictate how a protein folds in an aqueous environment. Hydrophobic residues tend to cluster in the protein's interior, away from water, while hydrophilic residues are often found on the protein's surface.
-
Protein Stability: Disulfide bonds between cysteine residues, hydrogen bonds, and ionic interactions between charged side chains all contribute to protein stability.
-
Protein-Ligand Interactions: The specific arrangement of amino acid side chains in a protein's binding site allows it to interact selectively with other molecules, such as substrates, inhibitors, or other proteins.
-
Enzyme Activity: The active sites of enzymes are precisely organized arrangements of amino acid side chains that facilitate catalytic reactions.
-
Protein-Protein Interactions: Many proteins function by interacting with other proteins. The specific amino acid residues involved in these interactions determine the specificity and strength of the interaction.
Post-translational Modifications: Expanding Amino Acid Diversity
After synthesis, proteins can undergo various modifications that alter the properties of their amino acid residues. These post-translational modifications (PTMs) include:
-
Phosphorylation: The addition of a phosphate group to serine, threonine, or tyrosine residues, often regulating protein activity.
-
Glycosylation: The addition of carbohydrate groups to asparagine, serine, or threonine residues, influencing protein stability, localization, and interactions.
-
Ubiquitination: The attachment of ubiquitin molecules, often targeting proteins for degradation.
-
Acetylation: The addition of an acetyl group, often modifying histone proteins and influencing gene expression.
These PTMs significantly expand the functional diversity of proteins, adding another layer of complexity to their roles in biological systems.
Conclusion: A Complex World of Protein Structure and Function
The 20 standard amino acids are the fundamental monomers that give rise to the incredible complexity and diversity of protein polymers. Their unique properties, along with the variations in amino acid sequence and post-translational modifications, contribute to the vast range of protein functions essential for life. Understanding the structure and properties of amino acids is paramount to comprehending the intricacies of protein biology and their crucial roles in all living organisms. Further research continually unveils new insights into the nuances of protein structure, function, and regulation, highlighting the ongoing importance of studying these remarkable biomolecules.
Latest Posts
Latest Posts
-
What Happens To An Animal Cell In A Isotonic Solution
Apr 04, 2025
-
Find A Cartesian Equation For The Curve
Apr 04, 2025
-
Abiotic Factor How To Get Water
Apr 04, 2025
-
Three Parts Of An Atp Molecule
Apr 04, 2025
-
What Unit Is Used To Measure Force
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about What Are The Monomers Of Protein Polymers . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
