In A Hypertonic Solution A Cell Will
listenit
Apr 02, 2025 · 5 min read
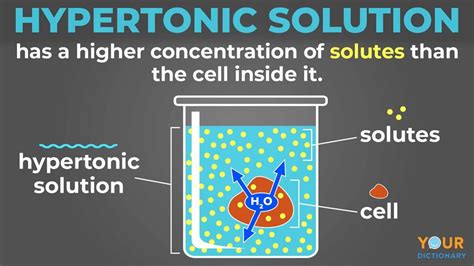
Table of Contents
In a Hypertonic Solution, a Cell Will: Shrinkage, Osmosis, and Cellular Responses
Understanding how cells behave in different environments is fundamental to biology. A crucial concept in this area is the impact of tonicity, specifically, what happens to a cell placed in a hypertonic solution. This article delves deep into the process, explaining the mechanisms involved, the consequences for the cell, and the diverse adaptations organisms have evolved to cope with hypertonic environments.
What is a Hypertonic Solution?
Before understanding cellular responses, we must define our terms. A hypertonic solution is one where the concentration of solutes (dissolved substances) is higher outside the cell than inside the cell. This difference in solute concentration creates an osmotic gradient, driving the movement of water.
Osmosis, a critical passive transport process, refers to the net movement of water across a selectively permeable membrane from a region of high water concentration (low solute concentration) to a region of low water concentration (high solute concentration). This movement continues until equilibrium is reached, or until a counteracting force prevents further water movement.
The Fate of a Cell in a Hypertonic Solution: Shrinkage
When a cell is immersed in a hypertonic solution, water follows the osmotic gradient, flowing out of the cell and into the surrounding solution. This leads to a decrease in the cell's volume and a resultant shrinkage, a phenomenon also known as plasmolysis in plant cells and crenation in animal cells.
Plasmolysis in Plant Cells
Plant cells possess a rigid cell wall that provides structural support. When a plant cell is placed in a hypertonic solution, water loss causes the cell membrane to pull away from the cell wall. This separation is plasmolysis. The cell becomes flaccid, losing its turgor pressure—the pressure exerted by the cell contents against the cell wall. Severe plasmolysis can damage the cell, potentially leading to cell death.
Crenation in Animal Cells
Animal cells lack a rigid cell wall. In a hypertonic solution, the water loss leads to the cell shrinking and becoming wrinkled. This process, crenation, can also disrupt cellular function and, if severe enough, cause cell death. The cell membrane may become damaged, compromising its integrity and potentially releasing cellular contents.
Mechanisms and Factors Affecting Cell Response
Several factors influence the extent and rate of shrinkage in a hypertonic solution:
1. The Magnitude of the Osmotic Gradient:
The greater the difference in solute concentration between the cell and the surrounding solution, the steeper the osmotic gradient, and the faster and more significant the water loss and shrinkage. A small difference might result in minor shrinkage, while a large difference could cause severe plasmolysis or crenation.
2. Cell Membrane Permeability:
The cell membrane's permeability to water and solutes plays a vital role. A more permeable membrane allows for a faster rate of water movement, accelerating the shrinkage process. Conversely, a less permeable membrane will slow down the water loss.
3. Surface Area to Volume Ratio:
Cells with a larger surface area relative to their volume will experience a faster rate of water loss compared to cells with a smaller surface area-to-volume ratio. This is because more membrane surface is exposed to the hypertonic solution.
4. Cell Wall Properties (Plant Cells):
In plant cells, the elasticity and strength of the cell wall influence the extent of plasmolysis. A stronger, less elastic cell wall will resist the inward pull of the membrane, reducing the extent of shrinkage compared to cells with weaker or more elastic walls.
Cellular Responses and Adaptations
Organisms inhabiting hypertonic environments have evolved diverse mechanisms to cope with the challenges posed by these conditions.
1. Osmoregulation:
Many organisms, especially those living in saltwater environments, employ sophisticated osmoregulatory mechanisms to control their internal water and solute balance. These mechanisms often involve specialized organs or cells that actively transport ions or other solutes to maintain a relatively constant internal osmotic pressure, preventing excessive water loss. Examples include specialized kidneys in mammals or contractile vacuoles in certain protists.
2. Accumulation of Compatible Solutes:
Some organisms accumulate compatible solutes within their cells. These are organic compounds that do not interfere with cellular metabolism, even at high concentrations. By increasing the internal solute concentration, they reduce the osmotic gradient and prevent excessive water loss. Examples of compatible solutes include various sugars, amino acids, and polyols.
3. Modification of Cell Wall Composition (Plant Cells):
Certain plants adapted to salty environments modify the composition of their cell walls to enhance their resilience to water stress. These modifications can help maintain cell turgor and prevent excessive plasmolysis even in hypertonic conditions.
4. Stress Response Pathways:
Exposure to hypertonic conditions triggers various stress response pathways within the cell. These pathways involve complex signaling cascades that activate genes encoding proteins involved in osmoprotection, repairing cellular damage, and maintaining cellular homeostasis.
Practical Applications and Significance
Understanding the effects of hypertonic solutions has several practical applications across various fields:
1. Food Preservation:
Hypertonic solutions, like concentrated sugar or salt solutions, are used in food preservation. The high solute concentration draws water out of microorganisms, inhibiting their growth and preventing spoilage.
2. Medicine:
Osmosis plays a critical role in various medical treatments. Intravenous fluids must have the correct tonicity to avoid damaging red blood cells. Hypertonic solutions can be used in certain medical procedures, such as reducing swelling.
3. Agriculture:
Understanding how plants respond to hypertonic conditions is crucial in agriculture, particularly in arid and saline regions. Developing salt-tolerant crops is an essential aspect of ensuring food security in such areas.
4. Biotechnology:
In biotechnology, controlling the tonicity of solutions is vital for culturing cells and tissues in vitro. The proper osmotic environment is essential for cell viability and function.
Conclusion
The response of a cell to a hypertonic solution, whether shrinkage through plasmolysis or crenation, is a fundamental biological phenomenon driven by osmosis. The extent of shrinkage depends on several factors, including the osmotic gradient, cell membrane permeability, and cellular adaptations. Organisms have evolved diverse mechanisms to cope with hypertonic environments, highlighting the crucial role of osmoregulation in maintaining cellular homeostasis and survival. The understanding of these processes has far-reaching implications in various fields, from food preservation to medicine and biotechnology. Further research into cellular responses to osmotic stress will continue to enhance our knowledge and enable the development of new strategies for mitigating the negative effects of hypertonic conditions.
Latest Posts
Latest Posts
-
What Are The Monomers Of Protein Polymers
Apr 03, 2025
-
How To Find Relative Maxima And Minima
Apr 03, 2025
-
How Do I Convert 3 8 Into A Decimal
Apr 03, 2025
-
What Is The Value Of H 2
Apr 03, 2025
-
What Is The Common Factor Of 8 And 12
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about In A Hypertonic Solution A Cell Will . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
