How Many Electrons Does Mn Have
listenit
Apr 05, 2025 · 5 min read
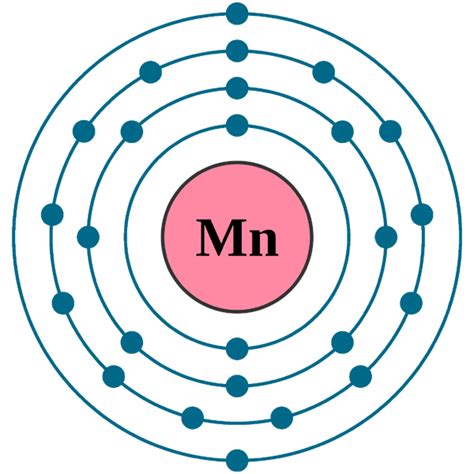
Table of Contents
How Many Electrons Does Mn Have? Unveiling the Secrets of Manganese
Manganese (Mn), a fascinating transition metal, plays a crucial role in various biological and industrial processes. Understanding its atomic structure, particularly the number of electrons it possesses, is key to comprehending its properties and behavior. This comprehensive guide delves deep into the electron configuration of manganese, exploring its implications and applications.
Understanding Atomic Structure: Protons, Neutrons, and Electrons
Before we dive into the specifics of manganese's electron count, let's establish a foundational understanding of atomic structure. Every atom consists of three fundamental subatomic particles:
- Protons: Positively charged particles residing in the atom's nucleus. The number of protons defines the element's atomic number and its identity.
- Neutrons: Neutrally charged particles also found in the nucleus. The number of neutrons can vary within an element, leading to isotopes.
- Electrons: Negatively charged particles orbiting the nucleus in specific energy levels or shells. The number of electrons typically equals the number of protons in a neutral atom.
Manganese's Atomic Number and Electron Configuration
Manganese's atomic number is 25, meaning a neutral manganese atom contains 25 protons. Since a neutral atom has an equal number of protons and electrons, a neutral manganese atom also possesses 25 electrons.
Now, let's explore how these 25 electrons are arranged within the atom's electron shells. Electrons occupy shells according to the Aufbau principle, filling lower energy levels before moving to higher ones. This arrangement dictates the atom's chemical properties and reactivity.
The electron configuration of manganese is: 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d⁵. Let's break this down:
- 1s²: The first energy level (shell) holds a maximum of two electrons, both in the 's' subshell.
- 2s² 2p⁶: The second energy level holds a total of eight electrons – two in the 's' subshell and six in the 'p' subshell.
- 3s² 3p⁶: The third energy level also holds eight electrons, mirroring the second level's arrangement.
- 4s² 3d⁵: This is where things get interesting. The fourth energy level begins to fill, with two electrons occupying the 's' subshell. However, the 3d subshell, which is slightly higher in energy than the 4s, also starts filling, containing five electrons in manganese.
This specific electron configuration is responsible for manganese's unique properties and its ability to form various oxidation states.
Manganese's Variable Oxidation States: A Consequence of Electron Configuration
The presence of five electrons in the 3d subshell and two electrons in the 4s subshell allows manganese to exhibit a wide range of oxidation states, meaning it can lose varying numbers of electrons to form ions. Common oxidation states of manganese include:
- +2 (Mn²⁺): Manganese loses two electrons, typically from the 4s subshell.
- +3 (Mn³⁺): Manganese loses three electrons, usually two from the 4s and one from the 3d subshell.
- +4 (Mn⁴⁺): Manganese loses four electrons.
- +5 (Mn⁵⁺): Manganese loses five electrons.
- +6 (Mn⁶⁺): Manganese loses six electrons.
- +7 (Mn⁷⁺): Manganese loses seven electrons. This is the highest oxidation state for manganese.
The ability to exhibit multiple oxidation states contributes significantly to manganese's diverse chemical behavior and its involvement in various redox reactions.
The Importance of Manganese in Biological Systems
Manganese is an essential trace element for numerous life forms, playing crucial roles in various biological processes. Its presence in enzymes and other biological molecules is vital for:
- Photosynthesis: Manganese is a key component of the oxygen-evolving complex in plants, facilitating the production of oxygen during photosynthesis. The precise mechanism involves intricate electron transfer processes, directly related to manganese's electron configuration and oxidation state changes.
- Enzyme Activity: Manganese acts as a cofactor in several enzymes, influencing their catalytic activity. These enzymes are involved in various metabolic pathways, including carbohydrate metabolism and bone formation. The ability of manganese to readily accept and donate electrons makes it an ideal participant in these reactions.
- Bone Formation: Manganese contributes to the formation and maintenance of healthy bones. It's involved in the synthesis of glycosaminoglycans, crucial components of the extracellular matrix in bone tissue.
- Immune Function: Manganese plays a role in supporting a healthy immune system, although the exact mechanisms are still under investigation.
Manganese deficiency can lead to several health issues, highlighting the element's critical importance in maintaining overall well-being.
Industrial Applications of Manganese
Beyond its biological significance, manganese finds widespread applications in various industries:
- Steel Production: Manganese is a crucial alloying element in steel production, improving its strength, hardness, and toughness. Its addition enhances steel's machinability and resistance to wear and tear. The unique properties stemming from its electron configuration contribute directly to these improvements.
- Aluminum Alloys: Manganese is also used in aluminum alloys to enhance their strength and corrosion resistance.
- Batteries: Manganese dioxide (MnO₂) is a vital component in many battery systems, including alkaline batteries and dry-cell batteries. Its ability to undergo redox reactions, directly linked to its variable oxidation states, is key to its functionality in these applications.
- Pigments and Dyes: Manganese compounds are used as pigments and dyes, providing vibrant colors in various applications, from paints and coatings to ceramics and plastics.
Isotopes of Manganese: Variations in Neutron Count
While the number of electrons in a neutral manganese atom remains constant at 25, the number of neutrons can vary, leading to different isotopes of manganese. The most common isotope is manganese-55 (⁵⁵Mn), with 25 protons and 30 neutrons. Other isotopes exist, but they are generally less stable and radioactive. These variations in neutron count don't alter the electron configuration or the fundamental chemical properties of manganese, but they do affect its nuclear properties and stability.
Conclusion: The Significance of Manganese's 25 Electrons
The seemingly simple answer – manganese has 25 electrons – unlocks a wealth of information about this fascinating element. Its electron configuration dictates its variable oxidation states, driving its diverse chemical behavior and enabling its crucial roles in both biological systems and industrial processes. From its participation in photosynthesis and enzyme activity to its applications in steel production and battery technology, manganese's impact is extensive and underscores the importance of understanding its atomic structure and properties at a fundamental level. The 25 electrons are not merely a number; they are the key to understanding manganese's multifaceted nature and its profound significance in our world. Further research into manganese's intricate properties continues to unveil new insights and potential applications, highlighting the ongoing relevance of this essential element.
Latest Posts
Latest Posts
-
Find The Zeros Of A Function Algebraically
Apr 05, 2025
-
Does Ethylene Glycol Have Ion Dipole Forces
Apr 05, 2025
-
What Is Freezing In Celsius Degrees
Apr 05, 2025
-
What Is The Lcm Of 2 And 4
Apr 05, 2025
-
How To Get Rid Of Fractions
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about How Many Electrons Does Mn Have . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
