What Is Freezing In Celsius Degrees
listenit
Apr 05, 2025 · 6 min read
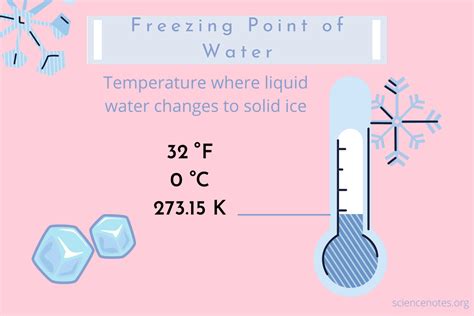
Table of Contents
What is Freezing in Celsius Degrees? A Deep Dive into Water's Phase Transition
Freezing, in the context of Celsius degrees, refers to the phase transition of water from its liquid state to its solid state, ice. This crucial point occurs at exactly 0°C (zero degrees Celsius) under standard atmospheric pressure (1 atmosphere or 101.325 kPa). This seemingly simple concept holds immense significance in various fields, from everyday life to complex scientific research. This article delves deep into the intricacies of freezing in Celsius, exploring its underlying principles, practical applications, and the factors influencing this fundamental process.
Understanding the Phase Transition of Water
The freezing point of water, 0°C, represents the temperature at which the kinetic energy of water molecules is reduced to a point where they can no longer overcome the attractive forces holding them in a liquid state. As the temperature drops below 0°C, these molecules lose enough energy to form a stable, ordered crystalline structure – ice. This process is exothermic, meaning it releases heat into the surroundings. The energy released is the latent heat of fusion, representing the energy needed to break the bonds holding the water molecules together in the solid state.
The Role of Molecular Structure
Water's unique molecular structure plays a vital role in its freezing behavior. Each water molecule (H₂O) consists of two hydrogen atoms covalently bonded to an oxygen atom. The oxygen atom's higher electronegativity leads to a polar molecule with a slightly positive end (hydrogen) and a slightly negative end (oxygen). This polarity results in hydrogen bonds—strong intermolecular forces—between water molecules. These bonds are responsible for water's high boiling point, surface tension, and, importantly, its unique freezing behavior.
When water freezes, the hydrogen bonds arrange the molecules into a regular, crystalline lattice structure. This lattice structure is less dense than the liquid state, which is why ice floats on water – a phenomenon crucial for aquatic life survival in colder climates. This lower density is due to the open hexagonal structure of ice crystals, leaving empty spaces within the lattice.
Factors Affecting the Freezing Point of Water
While 0°C is the standard freezing point of water, various factors can influence this temperature, subtly altering the phase transition. Understanding these factors is critical in many applications, from food preservation to industrial processes.
Pressure: The Pressure-Melting Point
Pressure significantly impacts the freezing point. Increasing pressure lowers the freezing point of water. This phenomenon, known as pressure-melting point, occurs because increasing pressure forces water molecules closer together, counteracting the tendency to form the less dense ice structure. This effect is relatively small at pressures close to atmospheric pressure, but it becomes more pronounced at higher pressures. This principle is utilized in ice skating, where the pressure from the skate blades momentarily melts the ice, providing a lubricating layer for smooth gliding.
Dissolved Substances: Freezing Point Depression
Adding dissolved substances (solutes) to water lowers its freezing point. This phenomenon, known as freezing point depression, is a colligative property, meaning it depends on the concentration of solute particles rather than their identity. The more solute particles present, the greater the freezing point depression. This principle is exploited in various applications such as de-icing roads (using salt) and creating antifreeze solutions for car radiators. The solutes interfere with the formation of the ice crystal lattice, hindering the freezing process and requiring a lower temperature to initiate it.
Impurities: The Role of Contamination
The presence of impurities in water, such as minerals or other dissolved substances, can also slightly alter the freezing point. Even seemingly pure water contains trace impurities that can affect the nucleation process – the initial formation of ice crystals. The presence of impurities can either accelerate or retard the freezing process, depending on their nature and concentration.
Supercooling: Delaying the Inevitable
Under specific conditions, water can remain in a liquid state even below 0°C. This phenomenon, known as supercooling, occurs when there are few nucleation sites – points where ice crystals can begin to form. In the absence of these sites, the water can exist in a metastable state, remaining liquid until disturbed or a nucleation site is introduced. A slight vibration or the introduction of an ice crystal can trigger rapid crystallization.
Applications of Freezing in Celsius
The freezing point of water at 0°C underpins numerous applications across various disciplines.
Food Preservation: Freezing as a Conservation Method
Freezing food is a widespread method of preservation based on slowing down or halting microbial growth and enzymatic activity. Lowering the temperature below 0°C forms ice crystals within the food, reducing the water activity available for microbial growth and enzymatic reactions. However, the formation of large ice crystals can damage food cell structure, impacting texture upon thawing. Therefore, rapid freezing techniques are often employed to minimize this damage.
Medicine and Biology: Cryopreservation and Cryogenics
Freezing plays a vital role in medicine and biology through cryopreservation techniques, used to preserve biological samples like cells, tissues, and organs. Controlled freezing processes are employed to minimize ice crystal formation damage and ensure the viability of samples after thawing. Cryogenics, involving extremely low temperatures, uses freezing to store and preserve materials for extended periods.
Civil Engineering and Construction: Dealing with Frost and Ice
Understanding freezing and its effects on water is crucial in civil engineering and construction. Frost action, where water in porous materials freezes and expands, can cause significant damage to structures. Engineers employ various techniques, such as frost-resistant materials and drainage systems, to mitigate these effects. De-icing roads and runways involves lowering the freezing point of water using substances like salt to prevent ice formation.
Chemistry and Materials Science: Studying Phase Transitions
The freezing of water serves as a model system for studying phase transitions in chemistry and materials science. Researchers investigate the thermodynamics and kinetics of freezing, seeking to understand the fundamental principles governing phase changes in various materials.
Environmental Science: Understanding Climate and Weather
The freezing point of water is fundamental to understanding climate and weather patterns. Freezing temperatures drive processes such as snow and ice formation, impacting hydrological cycles and ecosystems. Changes in freezing temperatures, as observed in global warming, have significant ecological and societal implications.
Beyond the Basics: Exploring Anomalies and Advanced Concepts
While 0°C represents the standard freezing point of water, there are nuances and exceptions to consider.
The Mpemba Effect: A Paradoxical Phenomenon
The Mpemba effect is a counterintuitive phenomenon where hot water freezes faster than cold water under certain conditions. Although not fully understood, various explanations involving evaporation, convection, and dissolved gases have been proposed. While not universally reproducible, the Mpemba effect highlights the complexities of the freezing process and the interplay of different factors influencing it.
Conclusion: The Ubiquity of Freezing at 0°C
Freezing at 0°C in Celsius is more than just a simple phase transition; it is a fundamental process with far-reaching implications across various scientific disciplines and everyday life. Understanding the intricacies of this process, from the role of molecular structure and hydrogen bonding to the influence of pressure, dissolved substances, and impurities, is essential for numerous applications. The ongoing research into freezing phenomena, including anomalies like the Mpemba effect, underscores the continuous pursuit of knowledge in this seemingly simple yet remarkably complex area of science. The ability to control and manipulate freezing processes has profound implications for food preservation, medicine, engineering, and our understanding of the natural world. From the everyday act of freezing water for a refreshing drink to sophisticated cryogenic preservation techniques, the significance of 0°C in Celsius remains undeniable and pervasive.
Latest Posts
Latest Posts
-
A 20 Foot Ladder Is Set Up Against A Building
Apr 06, 2025
-
How Do You Do Shorthand Electron Configuration
Apr 06, 2025
-
Three Less Than Six Times A Number
Apr 06, 2025
-
Mass Of Hydrogen Atom In G
Apr 06, 2025
-
What Is 240 Inches In Feet
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about What Is Freezing In Celsius Degrees . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
