Does Ethylene Glycol Have Ion Dipole Forces
listenit
Apr 05, 2025 · 5 min read
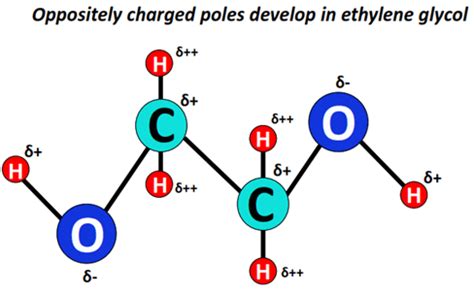
Table of Contents
Does Ethylene Glycol Have Ion-Dipole Forces? Understanding Intermolecular Forces in Ethylene Glycol
Ethylene glycol, a common ingredient in antifreeze and other industrial applications, possesses a fascinating array of intermolecular forces that govern its physical and chemical properties. Understanding these forces is crucial for predicting its behavior in various contexts. One frequently asked question regarding ethylene glycol's intermolecular interactions revolves around the presence of ion-dipole forces. This comprehensive article delves deep into the molecular structure of ethylene glycol and analyzes whether ion-dipole forces play a significant role in its interactions. We'll explore the dominant intermolecular forces present and explain why certain forces are more prevalent than others.
Understanding Intermolecular Forces
Before focusing specifically on ethylene glycol, let's establish a clear understanding of the various types of intermolecular forces. These forces are crucial in determining a substance's physical properties, such as boiling point, melting point, viscosity, and solubility. The key intermolecular forces include:
-
London Dispersion Forces (LDFs): These are the weakest intermolecular forces and are present in all molecules, regardless of their polarity. They arise from temporary fluctuations in electron distribution, creating temporary dipoles that induce dipoles in neighboring molecules. The strength of LDFs increases with the size and shape of the molecule.
-
Dipole-Dipole Forces: These forces occur between polar molecules, meaning molecules with a permanent dipole moment due to an uneven distribution of electron density. The positive end of one molecule attracts the negative end of another, resulting in a stronger attraction than LDFs.
-
Hydrogen Bonding: A special type of dipole-dipole interaction, hydrogen bonding occurs when a hydrogen atom bonded to a highly electronegative atom (like oxygen, nitrogen, or fluorine) is attracted to a lone pair of electrons on another electronegative atom in a nearby molecule. Hydrogen bonds are significantly stronger than typical dipole-dipole forces.
-
Ion-Dipole Forces: These forces arise from the interaction between an ion (either a cation or an anion) and a polar molecule. The charged ion is attracted to the oppositely charged end of the polar molecule. This is a relatively strong intermolecular force.
The Molecular Structure of Ethylene Glycol
Ethylene glycol (C₂H₆O₂) has the chemical formula HOCH₂CH₂OH. Its structure consists of two hydroxyl (-OH) groups attached to adjacent carbon atoms. This arrangement is crucial in determining its intermolecular forces. The hydroxyl groups are polar due to the significant electronegativity difference between oxygen and hydrogen. This polarity leads to several crucial implications.
Does Ethylene Glycol Exhibit Ion-Dipole Forces?
The answer is no, ethylene glycol does not exhibit significant ion-dipole forces under typical conditions. The reason is straightforward: ethylene glycol itself does not readily form ions. Ion-dipole forces require the presence of a free ion (a charged species) to interact with the polar molecule. Ethylene glycol is a neutral molecule; it doesn't readily dissociate into ions in solution.
While it's theoretically possible to generate ethylene glycol ions under extreme conditions (such as very strong acid or base reactions), this is not the typical state of ethylene glycol in most applications. In its usual form, as a liquid or dissolved in a solvent, it primarily exhibits other types of intermolecular forces.
Dominant Intermolecular Forces in Ethylene Glycol
Given the absence of ion-dipole forces, let's identify the dominant forces at play in ethylene glycol:
-
Hydrogen Bonding: The presence of two hydroxyl (-OH) groups makes hydrogen bonding the most significant intermolecular force in ethylene glycol. Each hydroxyl group can both donate and accept hydrogen bonds, leading to a strong network of intermolecular interactions. This explains ethylene glycol's relatively high boiling point (197 °C) compared to molecules of similar molecular weight that lack hydrogen bonding.
-
Dipole-Dipole Forces: Beyond hydrogen bonding, the overall polarity of the molecule contributes to dipole-dipole forces between ethylene glycol molecules. These forces, while weaker than hydrogen bonding, still contribute to the overall intermolecular attraction.
-
London Dispersion Forces: As with all molecules, London Dispersion Forces are present in ethylene glycol. However, their contribution is relatively minor compared to the much stronger hydrogen bonding and dipole-dipole interactions.
Implications of the Intermolecular Forces
The prevalence of hydrogen bonding and dipole-dipole forces in ethylene glycol significantly impacts its properties:
-
High Boiling Point: The strong hydrogen bonding network requires considerable energy to overcome, resulting in a relatively high boiling point.
-
High Viscosity: The strong intermolecular forces contribute to ethylene glycol's high viscosity, meaning it flows more slowly than many other liquids.
-
Solubility: Ethylene glycol is highly soluble in water due to the ability of its hydroxyl groups to form hydrogen bonds with water molecules. This makes it an excellent solvent for many polar substances.
-
Antifreeze Properties: The strong intermolecular interactions between ethylene glycol molecules and water molecules disrupt the formation of ice crystals, lowering the freezing point of water. This is the basis of its use as an antifreeze agent.
Differentiating Ethylene Glycol's Intermolecular Forces from Other Molecules
It's helpful to compare ethylene glycol's intermolecular forces to those of similar molecules to highlight the significance of hydrogen bonding:
-
Ethanol (CH₃CH₂OH): Ethanol also exhibits hydrogen bonding, but only one hydroxyl group per molecule means weaker overall intermolecular forces compared to ethylene glycol, leading to a lower boiling point.
-
Diethyl ether (CH₃CH₂OCH₂CH₃): Diethyl ether, while polar, lacks the hydroxyl groups necessary for hydrogen bonding. Its dominant intermolecular forces are dipole-dipole and London Dispersion Forces, resulting in a significantly lower boiling point than ethylene glycol.
-
Ethane (CH₃CH₃): Ethane is a nonpolar molecule, relying solely on London Dispersion Forces for intermolecular attraction, leading to a very low boiling point.
Conclusion: The Importance of Hydrogen Bonding in Ethylene Glycol
In summary, while ethylene glycol does not exhibit significant ion-dipole forces under typical conditions, it displays a robust network of intermolecular interactions dominated by hydrogen bonding and supplemented by dipole-dipole forces and weaker London Dispersion Forces. The strong hydrogen bonding network is the key factor responsible for its high boiling point, high viscosity, excellent solubility in water, and its effective use as an antifreeze agent. Understanding the interplay of these forces is essential for predicting and manipulating ethylene glycol's behavior in various applications, ranging from industrial processes to automotive applications. Further research into specific conditions might explore the possibility of inducing ionic interactions, but under normal circumstances, hydrogen bonding reigns supreme in dictating the behavior of this important compound.
Latest Posts
Latest Posts
-
How Do You Do Shorthand Electron Configuration
Apr 06, 2025
-
Three Less Than Six Times A Number
Apr 06, 2025
-
Mass Of Hydrogen Atom In G
Apr 06, 2025
-
What Is 240 Inches In Feet
Apr 06, 2025
-
What Is An Isosceles Obtuse Triangle
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about Does Ethylene Glycol Have Ion Dipole Forces . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
