How Many Electrons Are In Bromine
listenit
Apr 04, 2025 · 6 min read
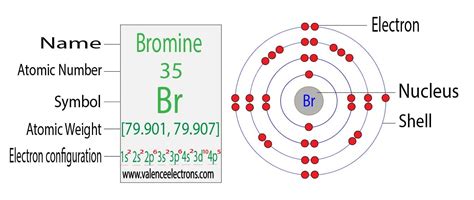
Table of Contents
How Many Electrons Are in Bromine? A Deep Dive into Atomic Structure
Bromine, a fascinating element with a rich history and unique properties, holds a special place in the periodic table. Understanding its atomic structure, particularly the number of electrons it possesses, is crucial to grasping its chemical behavior and its role in various applications. This comprehensive guide will not only answer the question, "How many electrons are in bromine?" but will also delve into the broader context of atomic structure, electron configuration, and the significance of electron count in determining an element's properties.
Understanding Atomic Structure: The Foundation of Bromine's Electron Count
Before we pinpoint the exact number of electrons in a bromine atom, let's establish a fundamental understanding of atomic structure. An atom is the basic building block of matter, composed of three primary subatomic particles:
- Protons: Positively charged particles located in the atom's nucleus. The number of protons defines the element's atomic number and its identity.
- Neutrons: Neutrally charged particles also residing in the nucleus. The number of neutrons can vary within an element, leading to isotopes.
- Electrons: Negatively charged particles orbiting the nucleus in specific energy levels or shells. These electrons are responsible for the chemical properties of an element.
The arrangement of electrons in these shells determines an atom's reactivity and how it interacts with other atoms to form molecules and compounds.
Determining the Number of Electrons in Bromine
The key to determining the number of electrons in any atom lies in its atomic number. This number, typically represented by the symbol 'Z', indicates the number of protons in the nucleus. In a neutral atom, the number of electrons equals the number of protons to maintain electrical neutrality.
Bromine's atomic number is 35. This means a neutral bromine atom contains 35 protons in its nucleus. Consequently, it also possesses 35 electrons orbiting that nucleus.
Isotopes and Electron Count: A Subtle Nuance
While the number of electrons in a neutral bromine atom is always 35, it's important to mention the concept of isotopes. Isotopes are atoms of the same element with the same number of protons but differing numbers of neutrons. This variation in neutron count affects the atom's mass but does not alter its electron count in a neutral state. Different bromine isotopes will all have 35 electrons.
Electron Configuration: Unveiling the Electron Arrangement
Knowing the total number of electrons is only half the story. The way these electrons are arranged in energy levels (also known as shells and subshells) is crucial for understanding an element's chemical behavior. This arrangement is described by the electron configuration.
Bromine's electron configuration is: 1s²2s²2p⁶3s²3p⁶4s²3d¹⁰4p⁵. Let's break this down:
- 1s²: Two electrons in the first energy level (n=1), in the s subshell.
- 2s²: Two electrons in the second energy level (n=2), in the s subshell.
- 2p⁶: Six electrons in the second energy level (n=2), in the p subshell.
- 3s²: Two electrons in the third energy level (n=3), in the s subshell.
- 3p⁶: Six electrons in the third energy level (n=3), in the p subshell.
- 4s²: Two electrons in the fourth energy level (n=4), in the s subshell.
- 3d¹⁰: Ten electrons in the third energy level (n=3), in the d subshell. Note that the 3d subshell fills after the 4s subshell.
- 4p⁵: Five electrons in the fourth energy level (n=4), in the p subshell.
This configuration explains bromine's chemical reactivity. The five electrons in the 4p subshell are valence electrons – the electrons involved in chemical bonding. This incomplete outermost shell makes bromine highly reactive, readily accepting an electron to achieve a stable octet (eight electrons) in its outermost shell.
Bromine's Properties and the Significance of its Electron Count
Bromine's properties are directly linked to its 35 electrons and their specific arrangement. Its position in Group 17 (halogens) of the periodic table highlights its tendency to gain an electron to form a stable negative ion (bromide ion, Br⁻). This characteristic is responsible for many of bromine's key features:
- Reactivity: Bromine is a highly reactive nonmetal, readily forming compounds with metals and other nonmetals. Its tendency to gain an electron drives its participation in various chemical reactions.
- Oxidizing Agent: Due to its high electronegativity (its ability to attract electrons), bromine acts as a strong oxidizing agent, meaning it readily accepts electrons from other substances.
- Physical State: At room temperature, bromine exists as a reddish-brown liquid, a unique property among halogens. This liquid state is influenced by the intermolecular forces arising from the electron distribution within the molecule.
- Applications: The reactivity and properties of bromine make it valuable in various applications, including:
- Production of flame retardants: Bromine compounds are incorporated into materials to increase their resistance to fire.
- Water purification: Bromine-based compounds are used as disinfectants for water treatment.
- Medical applications: Certain bromine compounds find use in medicine, for example, as sedatives.
- Agricultural chemicals: Bromine compounds are used in some pesticides and herbicides.
Beyond the Basics: Exploring Deeper into Bromine's Electronic Structure
The electron configuration provides a simplified model of electron distribution. A more accurate representation considers the quantum mechanical nature of electrons, utilizing concepts like orbitals and quantum numbers. Each electron is described by a unique set of four quantum numbers (principal quantum number, azimuthal quantum number, magnetic quantum number, and spin quantum number). These numbers specify the electron's energy level, subshell, orbital orientation, and spin.
Understanding these advanced concepts provides a more nuanced picture of bromine's electronic structure and helps explain the subtle differences in its behavior under various conditions. For example, the subtle energy differences between orbitals can affect the way bromine interacts with electromagnetic radiation, explaining its absorption and emission spectra.
Conclusion: The Significance of Electron Count in Chemistry
The simple answer to "How many electrons are in bromine?" is 35. However, understanding the significance of this number goes far beyond a simple count. The number of electrons, their arrangement in energy levels, and the resulting electron configuration dictate bromine's reactivity, its physical and chemical properties, and ultimately, its role in a vast array of applications. This exploration of bromine's atomic structure highlights the fundamental relationship between an element's electron count and its place in the wider world of chemistry. From its reactive nature to its practical uses, bromine's 35 electrons play a critical role in shaping its identity and importance. This in-depth look serves as a testament to the power of understanding atomic structure and its impact on the macroscopic world around us.
Latest Posts
Latest Posts
-
Rectangle That Is Not A Parallelogram
Apr 04, 2025
-
How Many Electrons Does Carbon Have Available For Chemical Bonding
Apr 04, 2025
-
What Is It Called When Everyone Plays Together
Apr 04, 2025
-
Y Is Inversely Proportional To X
Apr 04, 2025
-
Is Baking Bread A Chemical Or Physical Change
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about How Many Electrons Are In Bromine . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
