Why Are Ionic Substances Soluble In Water
listenit
Apr 03, 2025 · 6 min read
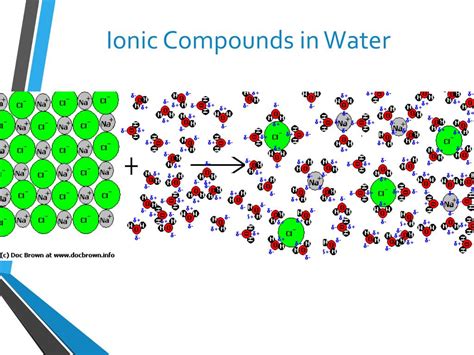
Table of Contents
Why Are Ionic Substances Soluble in Water? A Deep Dive into Polarity, Hydration, and Solubility
The solubility of ionic substances in water is a fundamental concept in chemistry with far-reaching implications across various fields, from biology and medicine to geology and engineering. Understanding why certain ionic compounds readily dissolve in water while others remain insoluble is crucial for predicting chemical behavior and designing effective processes. This comprehensive article delves into the intricacies of this phenomenon, explaining the underlying principles in detail.
The Role of Polarity: Water's Unique Structure
Water (H₂O) is a polar molecule. This means it possesses a partial positive charge (δ+) on the hydrogen atoms and a partial negative charge (δ−) on the oxygen atom. This charge separation arises from the difference in electronegativity between oxygen and hydrogen; oxygen is significantly more electronegative, attracting the shared electrons more strongly. This uneven distribution of charge creates a dipole moment, making water an excellent solvent for many ionic compounds.
Understanding Electronegativity and Dipole Moments
Electronegativity is a measure of an atom's ability to attract electrons in a chemical bond. A larger difference in electronegativity between two atoms leads to a more polar bond. The dipole moment, a vector quantity, represents the magnitude and direction of the charge separation in a molecule. Water's bent molecular geometry further enhances its polarity, resulting in a significant dipole moment.
The Ionic Bond: A Strong Electrostatic Attraction
Ionic substances, also known as salts, are formed through the electrostatic attraction between oppositely charged ions. These ions are formed when one atom loses electrons (becoming a positively charged cation) and another atom gains those electrons (becoming a negatively charged anion). This transfer of electrons creates a strong ionic bond, holding the ions together in a crystal lattice structure.
Crystal Lattice Structure and Lattice Energy
The crystal lattice is a highly ordered, three-dimensional arrangement of ions, maximizing electrostatic attraction while minimizing repulsion. The strength of the ionic bonds in the lattice is quantified by the lattice energy, which is the energy required to separate one mole of an ionic compound into its gaseous ions. High lattice energy indicates strong ionic bonds, making the compound less soluble.
The Hydration Process: Surrounding Ions with Water Molecules
The solubility of an ionic compound in water depends on the competition between the lattice energy (holding the ions together) and the hydration energy (the energy released when ions are surrounded by water molecules). The process of ions becoming surrounded by water molecules is called hydration.
How Hydration Works: Ion-Dipole Interactions
Water molecules, being polar, interact strongly with the charged ions. The partially negative oxygen atoms of water molecules are attracted to the positively charged cations, while the partially positive hydrogen atoms are attracted to the negatively charged anions. These interactions are called ion-dipole interactions. These interactions are strong enough to overcome the electrostatic attractions within the ionic lattice, leading to dissolution.
Hydration Energy: A Crucial Factor in Solubility
The hydration energy is the energy released when ions are hydrated. A high hydration energy favors solubility because it provides the energy needed to overcome the lattice energy. The magnitude of hydration energy depends on the charge density of the ions. Smaller, highly charged ions have higher charge densities and thus experience stronger ion-dipole interactions, resulting in higher hydration energies.
Factors Affecting Solubility of Ionic Compounds in Water
Several factors influence the solubility of ionic compounds in water beyond the simple interplay of lattice and hydration energies:
1. Charge Density of Ions: Size Matters
The charge density, defined as the charge of an ion divided by its size, is a crucial factor. Smaller ions with higher charges have higher charge densities, leading to stronger ion-dipole interactions and higher solubility. Conversely, larger ions with lower charges have lower charge densities and weaker interactions, resulting in lower solubility.
2. Temperature: The Effect of Kinetic Energy
Increasing the temperature increases the kinetic energy of both water molecules and ions. This increased kinetic energy helps overcome the attractive forces within the ionic lattice, promoting solubility. However, the effect of temperature on solubility is not always straightforward and can vary depending on the specific ionic compound.
3. Pressure: A Minor Influence on Solubility
Pressure has a relatively minor effect on the solubility of ionic compounds in water compared to temperature. The effect is more pronounced for gases dissolving in water.
4. Common Ion Effect: The Role of Equilibrium
The common ion effect states that the solubility of a sparingly soluble ionic compound decreases when a soluble salt containing a common ion is added to the solution. This is because the presence of the common ion shifts the solubility equilibrium, reducing the dissolution of the sparingly soluble salt.
Exceptions and Insoluble Ionic Compounds
While many ionic compounds are soluble in water, some are virtually insoluble. This occurs when the lattice energy is significantly greater than the hydration energy. The strong electrostatic attractions within the ionic lattice cannot be overcome by the ion-dipole interactions with water molecules. Examples include many metal sulfides, carbonates, and phosphates.
Predicting Solubility: Using Solubility Rules
Chemists use solubility rules to predict the solubility of ionic compounds in water. These rules are generalizations based on experimental observations and provide a useful framework for understanding solubility trends. However, it's crucial to remember that these are guidelines, and exceptions exist.
Applications of Understanding Ionic Solubility
The understanding of ionic solubility has numerous applications in various fields:
-
Medicine: Solubility is critical in designing drug delivery systems. Drugs must be soluble enough to be absorbed into the bloodstream but not so soluble that they are rapidly eliminated.
-
Environmental Science: Solubility plays a vital role in understanding the fate and transport of pollutants in the environment. The solubility of heavy metals, for instance, influences their bioavailability and toxicity.
-
Agriculture: Solubility of fertilizers determines how effectively plants absorb nutrients from the soil.
-
Industrial Processes: Many industrial processes rely on the solubility of ionic compounds, such as in the production of chemicals, the purification of water, and the extraction of minerals.
Conclusion: A Complex but Essential Phenomenon
The solubility of ionic substances in water is a complex phenomenon governed by the interplay of several factors, primarily the lattice energy and hydration energy. While the polar nature of water and the ion-dipole interactions are central to the process, factors like ion size, charge, temperature, and the presence of common ions can significantly influence solubility. Understanding these principles is crucial for a wide range of applications across various scientific and technological domains. Further research continues to refine our understanding of this fundamental aspect of chemistry, leading to innovative advancements in diverse fields. This detailed exploration provides a comprehensive overview, enabling a deeper grasp of this essential concept.
Latest Posts
Latest Posts
-
What Is The Square Root Of 67
Apr 04, 2025
-
What Is The Gcf Of 24 And 42
Apr 04, 2025
-
What Are The Natural Resources In The Northeast Region
Apr 04, 2025
-
What Is The Negative Square Root Of 64
Apr 04, 2025
-
Anything That Takes Up Space And Has Mass Is
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Why Are Ionic Substances Soluble In Water . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
