Which One Increases Number Of Collisions Between The Reactants
listenit
Apr 04, 2025 · 6 min read
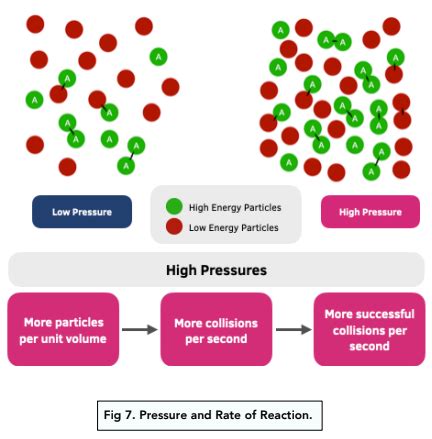
Table of Contents
Which Factor Increases the Number of Collisions Between Reactants? Maximizing Reaction Rates
Chemical reactions are all about collisions. The more collisions between reactant molecules, the faster the reaction proceeds. But not all collisions are created equal; only effective collisions, those possessing sufficient energy and correct orientation, lead to product formation. This article delves deep into the factors that influence the frequency of collisions between reactants, ultimately affecting the rate of a chemical reaction. Understanding these factors is crucial for optimizing chemical processes in various fields, from industrial manufacturing to biological systems.
The Fundamental Role of Collisions in Chemical Reactions
Before diving into specific factors, let's establish the fundamental principle: chemical reactions occur when reactant molecules collide with sufficient energy and proper orientation. This means the molecules must not only bump into each other but also possess enough kinetic energy to overcome the activation energy barrier – the minimum energy required for the reaction to proceed. The correct orientation ensures that the reactive parts of the molecules interact effectively.
The rate of a reaction is directly proportional to the number of effective collisions per unit time. Increasing the frequency of collisions generally increases the rate, even if the proportion of effective collisions remains constant. Therefore, manipulating factors that affect collision frequency is a key strategy for reaction rate control.
Factors Increasing the Number of Collisions
Several factors can significantly increase the number of collisions between reactant molecules. These include:
1. Concentration of Reactants:
This is perhaps the most intuitive factor. Increasing the concentration of reactants directly increases the number of molecules present in a given volume. With more molecules jostling around in the same space, the likelihood of collisions – both effective and ineffective – dramatically increases.
- Mechanism: Higher concentration means a higher molecular density. This leads to more frequent encounters between reactant molecules, increasing the overall collision rate.
- Example: Consider a reaction between two gases, A and B. If you double the concentration of A, you effectively double the number of A molecules available to collide with B molecules, thus doubling the collision frequency (assuming the concentration of B remains constant).
2. Temperature:
Temperature is a crucial factor influencing both the frequency and effectiveness of collisions. Higher temperatures lead to a higher average kinetic energy of the molecules.
- Mechanism: Increased kinetic energy translates to faster molecular movement. Faster molecules cover more distance in a given time, increasing the chance of collisions. Moreover, a higher temperature increases the proportion of molecules with kinetic energy exceeding the activation energy, resulting in a higher proportion of effective collisions.
- Example: Consider a reaction that proceeds slowly at room temperature. Increasing the temperature, say by 10°C, significantly accelerates the reaction due to the combined effect of increased collision frequency and a larger proportion of effective collisions. The increase in the rate constant, k, is typically exponential with temperature according to the Arrhenius equation.
3. Surface Area of Reactants:
This factor is particularly relevant for reactions involving solids. Increasing the surface area of a solid reactant increases the number of exposed molecules available for interaction with other reactants.
- Mechanism: A larger surface area provides more contact points for collisions. A finely powdered solid will react much faster than a single, large lump of the same solid because it offers a far greater surface area.
- Example: The reaction between a metal and an acid is significantly faster if the metal is in powder form compared to a solid block. The powdered metal presents a vastly larger surface area, leading to more frequent collisions between the metal atoms and acid molecules.
4. Pressure (for Gases):
For reactions involving gases, pressure plays a significant role. Increasing the pressure of a gaseous system increases the concentration of gas molecules.
- Mechanism: Higher pressure forces gas molecules closer together, increasing their density and, consequently, the collision frequency. This effect is similar to increasing concentration for liquid or solid reactants.
- Example: Reactions between gases proceed faster at higher pressures because the increased density leads to more frequent collisions between reactant molecules.
5. Stirring or Agitation:
Mechanical mixing or stirring enhances the rate of a reaction, especially in heterogeneous systems (those involving reactants in different phases). Stirring ensures reactants are evenly distributed and that fresh reactant molecules continuously reach the reaction zone.
- Mechanism: Stirring overcomes the diffusion limitations that could otherwise hinder the reactants from colliding. It essentially enhances the effective concentration of the reactants in the reaction zone.
- Example: In a reaction involving a solid and a liquid, stirring the mixture ensures that the solid particles are constantly exposed to fresh liquid reactants, maximizing the number of collisions. Without stirring, the reaction would be limited by the rate at which reactants diffuse into the reaction zone.
The Interplay of Factors: A Holistic Perspective
It's crucial to understand that these factors often work in conjunction. For instance, increasing the temperature of a gaseous reaction not only increases collision frequency due to increased kinetic energy but also increases the concentration (if the system is contained) due to thermal expansion. Similarly, increasing the surface area of a solid reactant in a liquid medium combined with stirring maximizes the reaction rate.
Optimizing reaction conditions involves carefully considering the interplay between these factors. A specific combination of temperature, pressure, concentration, surface area, and stirring might yield the fastest reaction rate for a given system.
Beyond Collision Frequency: The Importance of Effective Collisions
While increasing collision frequency significantly contributes to faster reaction rates, it's essential to remember that not all collisions are effective. Only collisions with sufficient energy and correct orientation lead to product formation.
Factors like temperature directly influence the proportion of effective collisions. Higher temperatures lead to a greater proportion of molecules with sufficient kinetic energy to overcome the activation energy barrier. Catalyst use can also impact this factor significantly. Catalysts provide an alternative reaction pathway that lowers the activation energy, allowing a greater proportion of collisions to be effective, even at lower temperatures.
Conclusion: A Multifaceted Approach to Reaction Rate Control
Increasing the number of collisions between reactant molecules is a fundamental strategy for enhancing reaction rates. This article explores several key factors that achieve this: concentration, temperature, surface area, pressure (for gases), and stirring. Optimizing reaction conditions involves a holistic consideration of these factors and their interplay, aiming to maximize both the frequency and effectiveness of collisions. Remember that while increasing collision frequency is crucial, the proportion of effective collisions, influenced by factors like temperature and catalysts, is equally important in determining the overall reaction rate. A deep understanding of these concepts is crucial for controlling chemical reactions in diverse applications, from industrial synthesis to biological processes.
Latest Posts
Latest Posts
-
Convert The Equation To Polar Form
Apr 04, 2025
-
Square Root Of 12 In Radical Form
Apr 04, 2025
-
What Is The Percentage Of 13 Out Of 16
Apr 04, 2025
-
How Many Kilograms Is 185 Pounds
Apr 04, 2025
-
What Is The Charge For Boron
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Which One Increases Number Of Collisions Between The Reactants . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
