What Is The Charge For Boron
listenit
Apr 04, 2025 · 5 min read
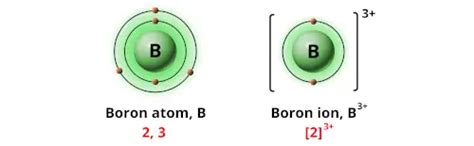
Table of Contents
What is the Charge of Boron? Understanding Boron's Oxidation States and Chemical Behavior
Boron, a fascinating metalloid element, occupies a unique position in the periodic table. Its behavior doesn't neatly align with either metals or nonmetals, leading to intriguing chemical properties and a range of oxidation states. Understanding the "charge" of boron, however, requires a nuanced approach beyond simply assigning a single numerical value. This article delves into the complexities of boron's electronic structure, its various oxidation states, and how these influence its chemical bonding and reactivity.
Boron's Electronic Structure: The Foundation of its Charge
Boron (B) has an atomic number of 5, meaning it possesses five protons and five electrons in its neutral state. Its electronic configuration is 1s²2s²2p¹, indicating that it has three valence electrons in its outermost shell (the second shell). These valence electrons are crucial in determining boron's bonding behavior and its potential charges. Unlike many other elements that readily gain or lose electrons to achieve a stable octet, boron exhibits a more complex and versatile approach to achieving electronic stability.
The Octet Rule and Boron's Exception
The octet rule, a fundamental principle in chemistry, states that atoms tend to gain, lose, or share electrons to achieve a stable configuration of eight electrons in their outermost shell. However, boron frequently forms compounds with only six electrons surrounding it, making it an exception to the octet rule. This is due to its relatively small size and the high energy required to accommodate additional electrons.
Boron's Oxidation States: A Spectrum of Charges
Instead of assigning a single "charge" to boron, it's more accurate to discuss its oxidation states. Oxidation state represents the hypothetical charge an atom would have if all bonds were completely ionic. While boron's bonds are often partially covalent, the oxidation state provides a useful framework for understanding its chemical behavior.
Boron exhibits several oxidation states, the most common being +3. This is because boron readily loses its three valence electrons to achieve a more stable electron configuration, resembling that of the noble gas helium. However, boron can also display other oxidation states, including:
-
+3: This is the most common and stable oxidation state for boron. Compounds in this oxidation state are numerous and well-studied. Examples include boric acid (H₃BO₃) and borax (Na₂B₄O₇·10H₂O).
-
+1: While less common than +3, boron can exhibit a +1 oxidation state in certain compounds. This occurs when only one or two of its valence electrons are involved in bonding. These compounds are often less stable than those with boron in the +3 oxidation state.
-
-1, -3: Under specific conditions, particularly in reactions with electropositive metals like alkali metals, boron can exhibit negative oxidation states. These are relatively rare and occur only in highly reducing environments.
Boron's Bonding Behavior: Covalent Bonds and Electron Deficiencies
Boron's bonding is predominantly covalent, meaning it shares electrons with other atoms to achieve a stable configuration. However, given its tendency to exhibit electron deficiencies (fewer than eight electrons in its valence shell), its covalent bonds are unique and often result in unusual structures.
Electron-Deficient Compounds: The Case of Boron Hydrides
Boron hydrides, compounds composed of boron and hydrogen, are excellent examples of electron-deficient compounds. They possess unusual structures with multicenter bonds, where electrons are shared among more than two atoms. These unique bonding characteristics are a direct consequence of boron's inability to achieve a complete octet easily. Examples include diborane (B₂H₆), a fascinating molecule with bridging hydrogen atoms.
Understanding the Charge in Different Chemical Contexts
The "charge" assigned to boron is highly context-dependent. In ionic compounds, it will be closer to +3. In covalent compounds, assigning a formal charge necessitates considering the electronegativity differences between boron and the atoms it bonds with. Formal charge provides an estimate of charge distribution within a molecule, but doesn't reflect the true, complex charge distribution present in a covalent bond.
Applications of Boron Compounds: Leveraging Diverse Oxidation States
Boron's diverse oxidation states and chemical properties contribute to its wide range of applications across various fields:
-
Glass and Ceramics: Boron oxide (B₂O₃) is a crucial ingredient in many types of glass, particularly borosilicate glass (Pyrex), known for its thermal resistance. Its inclusion modifies the glass's properties, making it less susceptible to thermal shock.
-
Semiconductors: Boron is a vital dopant in the semiconductor industry. Doping introduces impurities into silicon crystals, altering their electrical conductivity. Boron's ability to accept electrons makes it a p-type dopant, essential in creating semiconductor devices.
-
Nuclear Applications: Boron-10, an isotope of boron, is a significant neutron absorber. This property makes it useful in nuclear reactors as a control rod material and in radiation shielding.
-
Medicinal Applications: Boron compounds find application in medicine as well. Boron neutron capture therapy (BNCT) uses boron-10 to selectively target cancer cells.
-
Detergents and Cleaning Agents: Borax, a naturally occurring boron compound, is a common ingredient in laundry detergents and other cleaning agents due to its buffering and water-softening properties.
Conclusion: The Nuances of Boron's Charge
In conclusion, attributing a single "charge" to boron is an oversimplification. Its chemical behavior is best understood through the lens of its multiple oxidation states and the intricacies of its bonding. While the +3 oxidation state is prevalent, boron's capacity to exist in other oxidation states, coupled with its tendency to form electron-deficient compounds, renders it a fascinating and versatile element with a wide array of technological applications. Its behavior underscores the limitations of simple charge assignments and emphasizes the need to consider the complex interactions between electrons and atoms in understanding chemical behavior. This intricate nature contributes to boron's vital role in various industries and research fields. Further research into boron's chemical behavior continues to unveil new possibilities and applications for this unique element.
Latest Posts
Latest Posts
-
Why Must The Remainder Be Less Than The Divisor
Apr 04, 2025
-
How Many Inches In Two Yards
Apr 04, 2025
-
What Is 9 10 In Decimal Form
Apr 04, 2025
-
Rectangle That Is Not A Parallelogram
Apr 04, 2025
-
How Many Electrons Does Carbon Have Available For Chemical Bonding
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about What Is The Charge For Boron . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
