What Is The Proper Name For Mgf2
listenit
Apr 02, 2025 · 5 min read
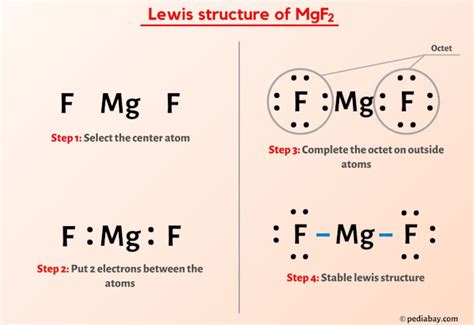
Table of Contents
What is the Proper Name for MgF2? A Deep Dive into Magnesium Fluoride
Magnesium fluoride (MgF2) is a crystalline solid with a range of applications, from optical coatings to specialized industrial uses. While its common name, magnesium fluoride, is widely accepted and understood, understanding its various names and chemical designations is crucial for clarity and accurate communication, especially in scientific and technical fields. This comprehensive guide will delve into the proper naming conventions for MgF2, exploring its chemical structure, properties, and the context in which different names might be used.
The Ubiquitous "Magnesium Fluoride"
The most common and universally accepted name for MgF2 is magnesium fluoride. This simple and straightforward nomenclature accurately reflects its chemical composition: magnesium (Mg) and fluoride (F2). It's the name you'll find in most textbooks, scientific publications, and commercial product descriptions. Its simplicity makes it ideal for broad communication and general understanding.
Beyond the Basics: Understanding Chemical Nomenclature
The simplicity of "magnesium fluoride" belies the complexity behind chemical nomenclature. While this common name is perfectly acceptable for most purposes, understanding the underlying system allows for precision and avoids ambiguity when dealing with more complex chemical compounds.
IUPAC Nomenclature
The International Union of Pure and Applied Chemistry (IUPAC) is the globally recognized authority on chemical nomenclature. IUPAC guidelines provide a systematic approach to naming chemical compounds, ensuring consistency and clarity across scientific disciplines. According to IUPAC nomenclature, MgF2 is correctly named magnesium difluoride. The prefix "di-" indicates the presence of two fluoride ions (F⁻) for every magnesium ion (Mg²⁺). While "magnesium fluoride" is widely accepted, "magnesium difluoride" is the more technically correct and precise IUPAC designation.
Other potential names
While less common, you might encounter other names for MgF2 in specialized contexts. These names usually emphasize specific properties or applications of the material:
- Fluorine magnesium: This alternative arrangement of the constituent elements is grammatically correct but less commonly used, and may even be confusing in some contexts.
- Magnesium difluoride crystal: This term is employed when referring to the crystalline structure of MgF2. This clarifies that we're discussing the material in its solid, crystalline form, as opposed to, say, a solution.
- MgF2 (chemical formula): The chemical formula itself serves as a precise designation, easily understood by anyone familiar with chemical notation. It is concise and unambiguous, especially in scientific contexts.
The Context Matters: Choosing the Right Name
The "proper" name for MgF2 ultimately depends on the context. While "magnesium difluoride" is technically more precise according to IUPAC guidelines, "magnesium fluoride" is perfectly acceptable and more widely understood in general contexts. Choosing the most appropriate name involves considering the audience and the purpose of the communication.
Examples:
- In a general science textbook: "Magnesium fluoride" is perfectly sufficient and readily understandable.
- In a research paper detailing the crystal structure: "Magnesium difluoride crystal" provides added clarity.
- In a chemical reaction equation: "MgF₂" (the chemical formula) is the most concise and unambiguous choice.
- In a commercial product description: "Magnesium fluoride" is easily understandable by customers without needing specialized chemical knowledge.
Exploring the Properties and Applications of MgF2
Understanding the properties of MgF2 is key to appreciating why it's chosen for various applications. It's a material with a remarkable combination of characteristics:
- High Transparency: MgF2 exhibits exceptional transparency across a wide range of wavelengths, from the ultraviolet (UV) to the infrared (IR) regions of the electromagnetic spectrum. This makes it crucial for optical components.
- Low Refractive Index: Compared to other optical materials, MgF2 possesses a relatively low refractive index. This is particularly beneficial in reducing reflection losses.
- Low Dispersion: This property signifies that the refractive index of MgF2 changes only slightly with changes in wavelength, leading to minimal chromatic aberration.
- Hardness and Durability: MgF2 is relatively hard and resistant to scratching and abrasion. This characteristic is valuable in applications where the material is subject to wear and tear.
- Chemical Stability: It demonstrates good chemical stability and resistance to corrosion. This quality contributes to its longevity in various applications.
- Low Solubility: Its low solubility in water makes it suitable for use in various environments without significant degradation.
These properties have made MgF2 invaluable in various applications:
- Optical Coatings: MgF2 is frequently used as an anti-reflection coating on lenses and other optical components. Its low refractive index effectively reduces unwanted reflections, improving image clarity and transmission efficiency.
- Optical Windows and Prisms: Its transparency and durability make it ideal for creating optical windows and prisms for use in lasers, spectrometers, and other optical instruments.
- Infrared Optics: The material's transparency extends into the infrared region, making it suitable for infrared imaging systems and other IR-related applications.
- UV Optics: MgF2’s UV transparency is crucial for applications in ultraviolet spectroscopy and lithography.
- Industrial Applications: MgF2 finds use in specialized industrial applications like high-temperature coatings and crucibles.
Conclusion: Precision and Clarity in Communication
While the most common and readily understandable name for MgF2 is "magnesium fluoride", the choice of the "proper" name is contextual. Using "magnesium difluoride" aligns with IUPAC recommendations and adds precision in technical settings. Understanding both names, along with the compound's chemical formula (MgF₂), enables clear and unambiguous communication across various contexts. The key is to choose the name that best serves the purpose and is most easily understood by the intended audience, ensuring effective communication and avoiding potential misunderstandings. Ultimately, the fundamental importance of MgF2 lies in its unique properties and wide-ranging applications, solidifying its position as a crucial material in diverse fields.
Latest Posts
Latest Posts
-
Which State Of Matter Takes The Shape Of Its Container
Apr 03, 2025
-
Whats The Square Root Of 145
Apr 03, 2025
-
What Is The Highest Common Factor Of 24 And 32
Apr 03, 2025
-
X 3y 9 In Slope Intercept Form
Apr 03, 2025
-
What State Of Matter Is Electricity
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about What Is The Proper Name For Mgf2 . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
