What 3 Particles Make Up An Atom
listenit
Apr 02, 2025 · 5 min read
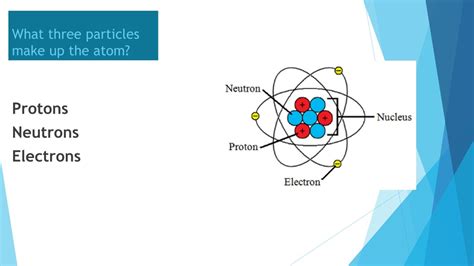
Table of Contents
What 3 Particles Make Up an Atom? A Deep Dive into Atomic Structure
The seemingly simple question, "What three particles make up an atom?" opens a door to a fascinating world of subatomic physics. While the answer – protons, neutrons, and electrons – is straightforward, a truly comprehensive understanding requires delving into the properties, behaviors, and significance of each particle within the atomic structure. This exploration goes beyond a simple definition, examining their roles in determining an element's properties and their implications in various scientific fields.
The Three Fundamental Subatomic Particles: Protons, Neutrons, and Electrons
Let's begin with a closer look at each of the three particles that constitute an atom:
1. Protons: The Positive Charge Carriers
Protons are positively charged subatomic particles found within the atom's nucleus. Their key characteristics include:
- Charge: +1 elementary charge (approximately 1.602 x 10<sup>-19</sup> Coulombs)
- Mass: Approximately 1.673 x 10<sup>-27</sup> kg (This is roughly 1836 times the mass of an electron)
- Location: Nucleus
- Symbol: p<sup>+</sup> or p
The number of protons in an atom's nucleus defines the element. This number is known as the atomic number. For example, hydrogen (H) has one proton (atomic number 1), helium (He) has two protons (atomic number 2), and so on. This fundamental property dictates the chemical behavior of an element. Changing the number of protons fundamentally alters the element itself.
2. Neutrons: The Neutral Nuclear Partners
Neutrons, as their name suggests, carry no electrical charge. They are also located within the atom's nucleus, alongside protons. Their properties include:
- Charge: 0
- Mass: Approximately 1.675 x 10<sup>-27</sup> kg (Slightly larger than a proton)
- Location: Nucleus
- Symbol: n<sup>0</sup> or n
Neutrons play a crucial role in stabilizing the atomic nucleus. The strong nuclear force, a fundamental force of nature, binds protons and neutrons together. Without neutrons, the electrostatic repulsion between positively charged protons would cause the nucleus to break apart, rendering stable atoms impossible. The number of neutrons in an atom can vary, leading to different isotopes of the same element. Isotopes have the same number of protons but a different number of neutrons. Some isotopes are stable, while others are radioactive.
3. Electrons: The Orbiting Negative Charge
Electrons are negatively charged particles that orbit the atom's nucleus. Unlike protons and neutrons, which are located within the nucleus, electrons reside in regions of space called electron shells or energy levels. Their properties are:
- Charge: -1 elementary charge (approximately -1.602 x 10<sup>-19</sup> Coulombs)
- Mass: Approximately 9.109 x 10<sup>-31</sup> kg (Significantly less massive than protons and neutrons)
- Location: Electron shells surrounding the nucleus
- Symbol: e<sup>-</sup> or e
The number of electrons in a neutral atom is equal to the number of protons. This balance of positive and negative charges results in a net charge of zero for the atom. However, atoms can gain or lose electrons, forming ions—charged atoms. When an atom loses electrons, it becomes a positively charged ion (cation), and when it gains electrons, it becomes a negatively charged ion (anion). The arrangement of electrons in an atom's shells determines its chemical reactivity and how it interacts with other atoms. This is the basis of chemical bonding and the formation of molecules.
Beyond the Basic Three: Exploring Subatomic Particle Complexity
While protons, neutrons, and electrons form the cornerstone of our understanding of atomic structure at a basic level, the reality is far more intricate. These particles themselves are composed of even smaller constituents called quarks and leptons.
Quarks: The Building Blocks of Protons and Neutrons
Protons and neutrons are not fundamental particles; they are composed of smaller particles called quarks. There are six types, or "flavors," of quarks: up, down, charm, strange, top, and bottom. Each quark carries a fractional electric charge.
- Protons: Consist of two up quarks (+2/3 charge each) and one down quark (-1/3 charge). The combined charge is +1.
- Neutrons: Consist of one up quark (+2/3 charge) and two down quarks (-1/3 charge each). The combined charge is 0.
The strong force, mediated by gluons (another type of elementary particle), holds quarks together within protons and neutrons.
Leptons: The Family of Electrons
Electrons belong to a class of fundamental particles called leptons. Leptons are fundamental particles that do not experience the strong force. Besides electrons, other leptons include muons and tau particles, each with their own associated neutrinos.
The Significance of Atomic Structure in Various Fields
The understanding of atomic structure is fundamental to numerous scientific fields:
1. Chemistry: Understanding Chemical Reactions
The arrangement of electrons in atoms determines how they interact with other atoms, forming chemical bonds. This knowledge is essential for understanding chemical reactions, predicting the properties of molecules, and developing new materials.
2. Physics: Exploring the Fundamental Forces
The study of atomic structure helps physicists explore the fundamental forces of nature—the strong force, the weak force, the electromagnetic force, and gravity—and their roles in governing the universe. Particle accelerators are used to probe the innermost workings of atoms and subatomic particles.
3. Nuclear Physics: Harnessing Nuclear Energy
Nuclear physics delves into the properties of atomic nuclei, including nuclear reactions like fission and fusion. This field is crucial for understanding nuclear energy, developing nuclear weapons, and utilizing radioisotopes in medicine and other applications.
4. Materials Science: Designing New Materials
An understanding of atomic structure is crucial for materials science, allowing scientists to tailor the properties of materials by manipulating their atomic arrangements. This knowledge is used to develop new materials with desired properties, such as strength, conductivity, or reactivity.
5. Astrophysics: Understanding Stellar Evolution
The behavior of atoms and their subatomic constituents plays a vital role in understanding stellar evolution, the formation of elements within stars, and the overall composition of the universe.
Conclusion: A Continuing Journey of Discovery
The simple answer to "What three particles make up an atom?"—protons, neutrons, and electrons—serves as a springboard for a deeper understanding of the universe's fundamental building blocks. From the intricate workings of quarks within protons and neutrons to the complex behavior of electrons in their shells, the study of atomic structure continues to reveal new insights into the nature of matter and energy. This journey of discovery continues to shape advancements across diverse fields, influencing technology, medicine, and our fundamental comprehension of the cosmos. The seemingly simple atom is, in reality, a microcosm of immense complexity and profound scientific importance.
Latest Posts
Latest Posts
-
Explain Why Chemical Equations Have To Be Balanced
Apr 03, 2025
-
How Do You Write 10 As A Fraction
Apr 03, 2025
-
Why Are Ionic Substances Soluble In Water
Apr 03, 2025
-
How Many Ml In A Dropper
Apr 03, 2025
-
How Many Bonds Does Boron Make
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about What 3 Particles Make Up An Atom . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
