The Basic Unit Of A Chemical Element
listenit
Apr 03, 2025 · 6 min read
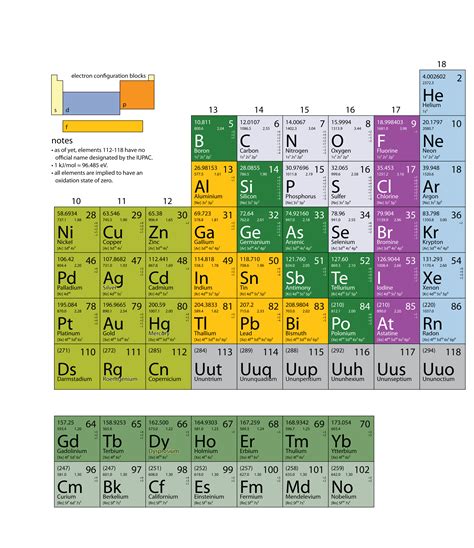
Table of Contents
The Basic Unit of a Chemical Element: Delving into the Atom
The fundamental building block of all matter, the very essence of what makes up everything around us, is the atom. Understanding the atom is crucial to grasping the complexities of chemistry, physics, and indeed, the entire universe. This article delves deep into the fascinating world of the atom, exploring its structure, properties, and its role as the basic unit of a chemical element.
What is an Atom?
An atom is the smallest unit of matter that retains the chemical properties of an element. Think of it as the indivisible particle – although, as we'll see, that indivisibility is somewhat of an oversimplification. Each atom is characterized by its atomic number, which represents the number of protons in its nucleus. This number uniquely defines the element. For example, an atom with one proton is hydrogen, an atom with two protons is helium, and so on.
Atoms are incredibly tiny. Their diameters are typically measured in angstroms (Å), where 1 Å = 10⁻¹⁰ meters. To put this into perspective, it would take about 10 billion atoms lined up to cover a centimeter! This minute size makes studying atoms incredibly challenging, requiring advanced techniques like electron microscopy and spectroscopy.
Key Components of an Atom:
The atom is comprised of three primary subatomic particles:
- Protons: These are positively charged particles found in the atom's nucleus. The number of protons determines the element's identity.
- Neutrons: These are neutral particles (no charge) also residing in the nucleus. They contribute to the atom's mass but not its charge. The number of neutrons can vary within the same element, resulting in isotopes.
- Electrons: These are negatively charged particles that orbit the nucleus in specific energy levels or shells. The number of electrons usually equals the number of protons in a neutral atom. Electron behavior dictates the atom's chemical properties and its interactions with other atoms.
The Atomic Nucleus: The Heart of the Atom
The nucleus, located at the center of the atom, is incredibly dense and contains almost all of the atom's mass. It's made up of protons and neutrons, collectively known as nucleons. The strong nuclear force, a powerful fundamental force, holds these nucleons together despite the electrostatic repulsion between the positively charged protons. The stability of the nucleus is a critical factor in determining an element's radioactivity.
Isotopes and their Significance:
Isotopes are atoms of the same element that have the same number of protons but a different number of neutrons. This means they have the same atomic number but different mass numbers (the sum of protons and neutrons). Many elements exist in nature as a mixture of isotopes. For example, carbon has three naturally occurring isotopes: carbon-12, carbon-13, and carbon-14. While they share the same chemical properties, their differing masses can impact their physical properties and have significant applications, particularly in fields like radiocarbon dating (using carbon-14).
Electron Shells and Energy Levels: The Atom's Outer Reaches
Electrons occupy specific energy levels or shells surrounding the nucleus. These shells are arranged in increasing distance from the nucleus, with each shell capable of holding a certain maximum number of electrons. The filling of these shells follows specific rules, dictated by quantum mechanics. The outermost shell, called the valence shell, contains the valence electrons, which play a crucial role in chemical bonding and reactivity.
Electron Configuration and Chemical Behavior:
The arrangement of electrons in different energy levels is known as the electron configuration. This configuration dictates the chemical behavior of an atom. Atoms tend to react in ways that achieve a stable electron configuration, often by gaining, losing, or sharing electrons with other atoms. This drive for stability underlies the formation of chemical bonds and the creation of molecules and compounds.
-
Noble Gases and Inertness: Noble gases, such as helium, neon, and argon, have completely filled valence shells. This makes them incredibly stable and unreactive, hence their inert nature.
-
Reactive Elements and Bonding: Elements with incomplete valence shells are more reactive. They tend to form chemical bonds with other atoms to achieve a stable electron configuration. These bonds can be ionic (transfer of electrons) or covalent (sharing of electrons).
Exploring the Quantum World: The Nature of Subatomic Particles
The behavior of electrons within an atom is not governed by classical physics but rather by the principles of quantum mechanics. This means that we cannot precisely know both the position and momentum of an electron simultaneously (Heisenberg's Uncertainty Principle). Instead, we describe electrons in terms of probabilities and wave functions.
Atomic Orbitals and Electron Clouds:
Quantum mechanics describes electron behavior using atomic orbitals, which are regions of space where there's a high probability of finding an electron. These orbitals are not fixed paths like planets orbiting a star; instead, they are more like fuzzy clouds of probability. The shape and orientation of these orbitals are determined by quantum numbers.
Beyond the Basic Atom: Ions and Molecules
Atoms can gain or lose electrons to form ions, which are charged particles. Cations are positively charged ions (lost electrons), while anions are negatively charged ions (gained electrons). This charge difference allows ions to interact electrostatically, forming ionic bonds. For example, the transfer of an electron from sodium (Na) to chlorine (Cl) forms the ionic compound sodium chloride (NaCl), or common table salt.
The Formation of Molecules:
Atoms can also share electrons to form covalent bonds, resulting in the creation of molecules. Molecules are groups of two or more atoms held together by chemical bonds. For example, two hydrogen atoms can share their electrons to form a hydrogen molecule (H₂). The properties of molecules differ significantly from the properties of the constituent atoms.
The Periodic Table: An Organization of Atoms
The periodic table organizes elements based on their atomic number and recurring chemical properties. The table's arrangement reflects the filling of electron shells and the resulting periodic trends in properties like electronegativity, ionization energy, and atomic radius. The periodic table is a powerful tool for understanding the relationships between elements and predicting their behavior.
Applications of Atomic Understanding
Our understanding of atoms has revolutionized numerous fields:
-
Medicine: Nuclear medicine uses radioactive isotopes for diagnosis and treatment. Medical imaging techniques, like MRI and PET scans, rely on principles of atomic physics.
-
Materials Science: By manipulating atomic structures, scientists create new materials with tailored properties for applications in various fields, including electronics, aerospace, and construction.
-
Energy Production: Nuclear power plants harness the energy released from nuclear reactions within atoms.
-
Technology: Microelectronics relies heavily on our understanding of atomic behavior to create ever-smaller and more powerful computer chips.
The Ongoing Exploration of the Atom:
Despite centuries of research, the atom remains a fascinating area of ongoing exploration. Scientists continue to push the boundaries of our understanding, probing deeper into the structure of the nucleus and investigating exotic forms of matter. New discoveries and technologies are constantly revealing further intricacies of the atomic world, leading to new advancements in science and technology. From the smallest subatomic particles to the largest molecules, the atom holds the key to understanding the fundamental nature of our reality. Its study continues to unlock countless possibilities and shape our future.
Latest Posts
Latest Posts
-
The Amount Of Energy Required To Raise The Temperature
Apr 04, 2025
-
38 Out Of 40 Is What Percent
Apr 04, 2025
-
Which Polynomial Represents The Sum Below
Apr 04, 2025
-
How Many Valence Electrons In Al
Apr 04, 2025
-
Silver Has A Density Of 10 5 G Cm3
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about The Basic Unit Of A Chemical Element . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
