How Many Valence Electrons In Al
listenit
Apr 04, 2025 · 6 min read
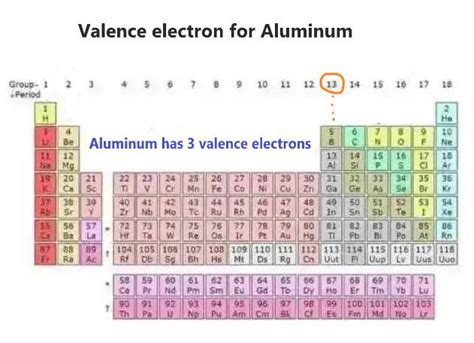
Table of Contents
How Many Valence Electrons Does Aluminum (Al) Have? A Deep Dive into Atomic Structure and Chemical Bonding
Aluminum, a ubiquitous metal found in everything from soda cans to aircraft parts, plays a crucial role in our modern world. Understanding its chemical behavior hinges on knowing its electronic structure, specifically the number of valence electrons. This article will delve deep into the answer to the question: How many valence electrons does aluminum have? We'll explore the concepts of electron shells, valence electrons, their significance in chemical bonding, and aluminum's unique properties stemming from its valence electron configuration.
Understanding Atomic Structure: Shells, Subshells, and Orbitals
Before diving into aluminum's valence electrons, let's refresh our understanding of atomic structure. Atoms are composed of a central nucleus containing protons and neutrons, surrounded by orbiting electrons. These electrons are arranged in distinct energy levels called shells. Each shell can hold a maximum number of electrons, determined by the formula 2n², where 'n' is the principal quantum number representing the shell's energy level (n=1, 2, 3, etc.).
Within each shell are subshells, denoted by the letters s, p, d, and f. These subshells have specific shapes and can hold a certain number of electrons:
- s subshell: Holds a maximum of 2 electrons.
- p subshell: Holds a maximum of 6 electrons.
- d subshell: Holds a maximum of 10 electrons.
- f subshell: Holds a maximum of 14 electrons.
Subshells are further divided into orbitals, which are regions of space where there's a high probability of finding an electron. Each orbital can hold a maximum of two electrons with opposite spins (Pauli Exclusion Principle).
Aluminum's Electronic Configuration: Unveiling the Valence Electrons
Aluminum (Al) has an atomic number of 13, meaning it has 13 protons and, in a neutral atom, 13 electrons. To determine the number of valence electrons, we need to understand how these electrons are distributed among the shells and subshells. This distribution is represented by the atom's electronic configuration.
Aluminum's electronic configuration is 1s²2s²2p⁶3s²3p¹. Let's break this down:
- 1s²: Two electrons occupy the 1s orbital in the first shell.
- 2s²: Two electrons occupy the 2s orbital in the second shell.
- 2p⁶: Six electrons occupy the three 2p orbitals in the second shell.
- 3s²: Two electrons occupy the 3s orbital in the third shell.
- 3p¹: One electron occupies one of the three 3p orbitals in the third shell.
The valence electrons are the electrons in the outermost shell, which for aluminum is the third shell. In aluminum's case, this includes the two electrons in the 3s orbital and the one electron in the 3p orbital.
Therefore, aluminum has 3 valence electrons.
Why are Valence Electrons Important?
Valence electrons are crucial because they determine an atom's chemical properties and how it interacts with other atoms. They are the electrons involved in chemical bonding, the process by which atoms combine to form molecules and compounds. Atoms tend to react in ways that achieve a stable electron configuration, often resembling the noble gases with filled outer shells (octet rule).
Aluminum's Chemical Behavior: A Consequence of Three Valence Electrons
Aluminum's three valence electrons explain its characteristic chemical behavior. It readily loses these three electrons to achieve a stable octet configuration, forming a +3 ion (Al³⁺). This tendency to lose electrons makes aluminum a highly reactive metal, particularly with oxidizing agents like oxygen.
Examples of Aluminum's Chemical Reactions:
-
Reaction with Oxygen: Aluminum readily reacts with oxygen in the air, forming a protective layer of aluminum oxide (Al₂O₃). This layer prevents further oxidation and accounts for aluminum's corrosion resistance. The reaction involves aluminum losing its three valence electrons to oxygen atoms.
-
Reaction with Acids: Aluminum reacts with strong acids, such as hydrochloric acid (HCl), releasing hydrogen gas and forming aluminum salts. Again, this reaction involves the loss of aluminum's three valence electrons.
-
Formation of Alloys: Aluminum's ability to readily form metallic bonds with other metals leads to the formation of numerous strong and lightweight alloys. These alloys find applications in various industries, including aerospace and automotive.
Aluminum's Properties and Applications: A Link to Valence Electrons
The unique properties of aluminum, responsible for its widespread applications, are directly linked to its three valence electrons and resulting chemical behavior:
-
Light Weight: Aluminum's low density, making it lighter than many other metals, is a consequence of its atomic structure and the relatively weak metallic bonding stemming from its three valence electrons.
-
High Strength-to-Weight Ratio: Despite its lightness, aluminum alloys exhibit high strength, making them ideal for structural applications. This is achieved through alloying with other metals that influence the strength of the metallic bonds.
-
Corrosion Resistance: The formation of a protective aluminum oxide layer significantly enhances aluminum's resistance to corrosion, further increasing its durability and longevity in various applications.
-
Excellent Conductivity: Aluminum's relatively high electrical and thermal conductivity are linked to its ability to easily conduct electrons through its metallic lattice. This property makes aluminum suitable for electrical wiring and heat exchangers.
Applications of Aluminum: A Diverse Landscape
The combination of properties stemming from its three valence electrons makes aluminum a versatile material with applications across numerous sectors:
-
Packaging: Aluminum foil and cans are ubiquitous in food and beverage packaging due to aluminum's malleability, corrosion resistance, and recyclability.
-
Transportation: Aluminum alloys are extensively used in aircraft manufacturing, automotive parts, and railway carriages due to their high strength-to-weight ratio.
-
Construction: Aluminum finds applications in building materials, including window frames, roofing, and cladding, due to its corrosion resistance and lightweight nature.
-
Electrical Engineering: Aluminum's high electrical conductivity makes it a popular choice for electrical wiring and transmission lines.
Beyond the Basics: Exploring Advanced Concepts Related to Aluminum's Valence Electrons
While the basic understanding of aluminum's three valence electrons provides a solid foundation for understanding its chemical behavior, more advanced concepts can offer a deeper insight:
-
Orbital Hybridization: In aluminum compounds, the 3s and 3p orbitals can hybridize to form new orbitals with different shapes and energies, impacting the geometry of the molecule. Understanding hybridization helps predict the molecular structure and properties of aluminum compounds.
-
Quantum Mechanics: A deeper understanding of aluminum's electron configuration requires the application of quantum mechanics, which provides a more accurate description of electron behavior and energy levels within the atom.
-
Computational Chemistry: Computational methods, utilizing sophisticated software and algorithms, can simulate and predict the properties of aluminum and its compounds, furthering our understanding of its behavior.
Conclusion: The Significance of Aluminum's Three Valence Electrons
The number of valence electrons an atom possesses is fundamental to its chemical and physical properties. Aluminum, with its three valence electrons, exhibits a characteristic reactivity, forming stable +3 ions and contributing to its unique properties. This understanding is crucial for comprehending its diverse applications across various industries. From the lightweight cans holding our beverages to the sturdy structures of airplanes, aluminum's properties, deeply rooted in its electronic structure and the behavior of its three valence electrons, continue to shape our world. Further exploration of the advanced concepts surrounding its electronic configuration promises even deeper insights into this remarkable element.
Latest Posts
Latest Posts
-
What Is The Purpose Of The Experiment
Apr 04, 2025
-
Why Must The Remainder Be Less Than The Divisor
Apr 04, 2025
-
How Many Inches In Two Yards
Apr 04, 2025
-
What Is 9 10 In Decimal Form
Apr 04, 2025
-
Rectangle That Is Not A Parallelogram
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about How Many Valence Electrons In Al . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
