Percent Composition Of Mg No3 2
listenit
Apr 02, 2025 · 5 min read
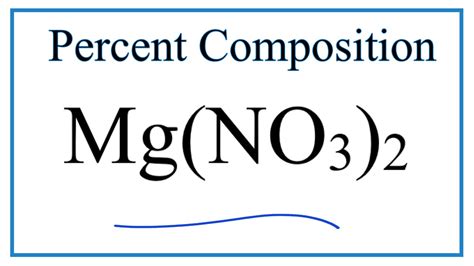
Table of Contents
Percent Composition of Mg(NO₃)₂: A Comprehensive Guide
Magnesium nitrate, Mg(NO₃)₂, is a common inorganic salt used in various applications, from fertilizers and pyrotechnics to its role in specialized chemical processes. Understanding its percent composition is crucial for many chemical calculations and applications. This comprehensive guide will delve into the detailed calculations and underlying concepts, equipping you with a thorough understanding of Mg(NO₃)₂'s elemental makeup.
Understanding Percent Composition
Percent composition, also known as percentage composition, represents the mass percentage of each element present in a compound. It's a fundamental concept in chemistry, essential for stoichiometry, empirical formula determination, and many other analytical techniques. The percent composition of a compound provides a concise way to express the relative amounts of each constituent element.
Calculating Percent Composition: The Formula
The fundamental formula for calculating the percent composition of an element in a compound is:
(Mass of the element in the compound / Molar mass of the compound) x 100%
This formula provides a straightforward method to determine the contribution of each element to the overall mass of the compound.
Determining the Percent Composition of Mg(NO₃)₂
To calculate the percent composition of Mg(NO₃)₂, we'll follow these steps:
Step 1: Find the Molar Mass of Each Element
First, we need the molar masses of each element present in Mg(NO₃)₂: Magnesium (Mg), Nitrogen (N), and Oxygen (O). These values can be found on the periodic table:
- Magnesium (Mg): 24.31 g/mol
- Nitrogen (N): 14.01 g/mol
- Oxygen (O): 16.00 g/mol
Step 2: Determine the Number of Atoms of Each Element
The chemical formula Mg(NO₃)₂ tells us the number of atoms of each element in one molecule of magnesium nitrate:
- Magnesium (Mg): 1 atom
- Nitrogen (N): 2 atoms (because of the subscript 2 outside the parenthesis)
- Oxygen (O): 6 atoms (2 from the subscript outside the parenthesis multiplied by 3 from the subscript of oxygen inside the parenthesis)
Step 3: Calculate the Total Mass of Each Element in One Mole of Mg(NO₃)₂
Now, we calculate the total mass of each element in one mole of Mg(NO₃)₂:
- Magnesium (Mg): 1 atom × 24.31 g/mol = 24.31 g
- Nitrogen (N): 2 atoms × 14.01 g/mol = 28.02 g
- Oxygen (O): 6 atoms × 16.00 g/mol = 96.00 g
Step 4: Calculate the Molar Mass of Mg(NO₃)₂
The molar mass of Mg(NO₃)₂ is the sum of the total mass of each element:
24.31 g + 28.02 g + 96.00 g = 148.33 g/mol
Step 5: Calculate the Percent Composition of Each Element
Finally, we use the formula to calculate the percent composition of each element:
-
Percent Composition of Magnesium (Mg): (24.31 g / 148.33 g) x 100% = 16.39%
-
Percent Composition of Nitrogen (N): (28.02 g / 148.33 g) x 100% = 18.88%
-
Percent Composition of Oxygen (O): (96.00 g / 148.33 g) x 100% = 64.73%
Verification of Results
To verify our calculations, we can check if the sum of the percent compositions adds up to approximately 100%. Any minor discrepancies are due to rounding errors:
16.39% + 18.88% + 64.73% = 100%
This confirms the accuracy of our calculations.
Applications of Percent Composition of Mg(NO₃)₂
Understanding the percent composition of Mg(NO₃)₂ has several practical applications:
-
Fertilizer Production: Knowing the percentage of nitrogen in Mg(NO₃)₂ helps determine the appropriate amount to use in fertilizers to provide the required nitrogen content for plant growth. Nitrogen is a crucial nutrient for plants.
-
Pyrotechnics: In pyrotechnics, Mg(NO₃)₂ acts as an oxidizer. Its composition determines its oxidizing power, affecting the intensity and duration of the pyrotechnic effect. Accurate composition is crucial for safety and predictable results.
-
Chemical Reactions: Percent composition is essential for stoichiometric calculations in chemical reactions involving Mg(NO₃)₂. Accurate calculations are needed for quantitative analysis and controlling reaction yields.
-
Analytical Chemistry: Percent composition is a fundamental parameter used in various analytical techniques, such as gravimetric analysis, to determine the purity and composition of samples containing Mg(NO₃)₂.
Advanced Concepts and Considerations
While the above calculations provide a basic understanding, several advanced concepts warrant consideration:
-
Isotopic Abundance: The molar masses used are average atomic weights, accounting for the natural isotopic abundance of each element. For highly precise calculations, the specific isotopic composition of the sample should be taken into account.
-
Hydrates: Mg(NO₃)₂ can exist as hydrates, meaning water molecules are incorporated into the crystal structure. The presence of water will alter the percent composition. The formula should specify whether it's an anhydrous salt or a specific hydrate (e.g., Mg(NO₃)₂·6H₂O).
-
Impurities: Real-world samples of Mg(NO₃)₂ might contain impurities. These impurities will affect the experimental percent composition, making it deviate from the theoretical value calculated above. Analytical methods are necessary to determine the purity of the sample before accurate percent composition can be determined.
Conclusion
Determining the percent composition of Mg(NO₃)₂ involves a straightforward yet crucial calculation in chemistry. This guide has provided a step-by-step approach, highlighting the importance of understanding molar masses, the number of atoms in the molecule, and the application of the percent composition formula. While the basic calculation is relatively simple, appreciating the advanced concepts and potential sources of error is essential for accurate and reliable results, particularly in practical applications where precise composition is critical. The knowledge gained can be applied to various chemical calculations and analytical methods, proving to be an invaluable tool for students and professionals in chemistry and related fields.
Latest Posts
Latest Posts
-
What Is The Least Common Multiple Of 20 And 30
Apr 03, 2025
-
What Is The Square Root Of 170
Apr 03, 2025
-
How Long Does A Sensory Memory Last
Apr 03, 2025
-
63 Is 90 Percent Of What
Apr 03, 2025
-
A Square Is Always A Parallelogram
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Percent Composition Of Mg No3 2 . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
