Lewis Structure For Co With Formal Charges
listenit
Apr 04, 2025 · 6 min read
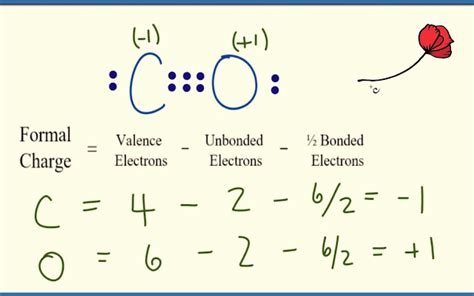
Table of Contents
Lewis Structure for CO with Formal Charges: A Deep Dive
Carbon monoxide (CO), a simple yet fascinating molecule, presents a unique challenge when it comes to drawing its Lewis structure and assigning formal charges. While seemingly straightforward, understanding the nuances of its bonding and the implications of formal charge distribution is crucial for comprehending its chemical behavior and reactivity. This article delves deep into the intricacies of constructing the Lewis structure for CO, meticulously explaining the process and the rationale behind assigning formal charges. We’ll also explore the significance of these formal charges in interpreting the molecule's properties.
Understanding Lewis Structures and Formal Charges
Before diving into the specifics of CO, let's briefly review the fundamental concepts of Lewis structures and formal charges.
Lewis structures, also known as electron dot diagrams, are visual representations of the valence electrons in a molecule. They illustrate how atoms share electrons to achieve a stable octet (or duet for hydrogen) configuration, mimicking the electronic configuration of noble gases. The structure includes chemical symbols for each atom, lines representing covalent bonds (shared electron pairs), and lone pairs (unshared electron pairs) represented by dots.
Formal charges are hypothetical charges assigned to atoms in a Lewis structure. They help to determine the most plausible structure when multiple resonance structures are possible. The formal charge is calculated as:
Formal Charge = (Valence electrons) - (Non-bonding electrons) - (½ Bonding electrons)
A lower magnitude of formal charges generally indicates a more stable Lewis structure. Ideally, a structure with formal charges closest to zero is preferred, although this isn't always achievable. The sum of formal charges in a neutral molecule should always equal zero.
Constructing the Lewis Structure for CO
Constructing the Lewis structure for CO involves several steps:
-
Counting Valence Electrons: Carbon has four valence electrons, and oxygen has six. Therefore, the total number of valence electrons in CO is 10.
-
Identifying the Central Atom: In most cases, the less electronegative atom acts as the central atom. However, in CO, both carbon and oxygen are relatively close in electronegativity. While oxygen is slightly more electronegative, it's more common to place carbon in the center, especially given the subsequent formal charge implications.
-
Connecting Atoms with Single Bonds: Connect the carbon and oxygen atoms with a single bond, which uses two valence electrons. This leaves eight electrons remaining.
-
Distributing Remaining Electrons as Lone Pairs: Place the remaining eight electrons as lone pairs around the oxygen atom. Oxygen can accommodate these lone pairs comfortably, fulfilling its octet. Carbon, however, only has two electrons around it at this stage.
-
Forming Multiple Bonds to Achieve Octet: To satisfy the octet rule for both atoms, we need to transform one or more lone pairs on oxygen into bonding pairs. Moving two lone pairs from oxygen to form two additional bonds with carbon will provide both atoms with a complete octet. This leads to the formation of a triple bond between carbon and oxygen.
-
The Final Lewis Structure: The resulting Lewis structure shows a triple bond (three shared electron pairs) between carbon and oxygen. Carbon has no lone pairs, and oxygen has two lone pairs.
Assigning Formal Charges to the CO Lewis Structure
Now, let's calculate the formal charges for each atom in the final Lewis structure:
For Carbon:
- Valence electrons = 4
- Non-bonding electrons = 0
- Bonding electrons = 6 (3 bonds x 2 electrons/bond)
- Formal Charge = 4 - 0 - (6/2) = +1
For Oxygen:
- Valence electrons = 6
- Non-bonding electrons = 4 (2 lone pairs x 2 electrons/lone pair)
- Bonding electrons = 6 (3 bonds x 2 electrons/bond)
- Formal Charge = 6 - 4 - (6/2) = -1
Therefore, the Lewis structure for CO with formal charges shows carbon with a formal charge of +1 and oxygen with a formal charge of -1.
Understanding the Implications of Formal Charges in CO
The presence of formal charges in the Lewis structure of CO reflects the molecule’s polarity. The positive formal charge on carbon and the negative formal charge on oxygen indicate that the electron density is shifted towards the oxygen atom, making oxygen partially negative (δ-) and carbon partially positive (δ+). This polarity is a significant factor in CO's reactivity and interactions with other molecules.
While the formal charges might seem to suggest a substantial ionic character, the triple bond creates significant covalent character. The strong bond between carbon and oxygen is a combination of covalent and polar covalent character. It's not simply a carbon cation and an oxygen anion; the electrons are shared, though unevenly.
Comparing CO to Other Molecules: Illustrating Formal Charge Significance
To further highlight the importance of formal charges, let's briefly compare CO to other molecules like CO2 and N2.
In CO2, the Lewis structure features two double bonds between carbon and each oxygen atom. Formal charges are zero for all atoms, indicating a more evenly distributed electron density compared to CO. This difference explains CO2's relatively lower polarity compared to CO.
In N2, the Lewis structure shows a triple bond between the two nitrogen atoms. Both nitrogen atoms have a formal charge of zero, indicating a nonpolar covalent bond. The absence of formal charges is consistent with N2's nonpolar nature.
Resonance Structures and Formal Charges in CO
Although the triple-bond structure is the most significant contributor to the overall bonding picture of CO, it's also crucial to acknowledge the potential for resonance. In principle, one could consider alternative resonance structures with different bond orders and formal charge distributions. However, the triple-bond structure with a +1 formal charge on carbon and a -1 formal charge on oxygen is the most energetically favourable and contributes most significantly to the overall molecular structure. Other resonance structures would be less stable due to a larger separation of charges and/or incomplete octets.
Beyond Formal Charges: A Deeper Look at Bonding in CO
While formal charges provide valuable insight, they are not the only factor that determines molecular properties. A complete understanding of CO's bonding requires considering:
- Molecular Orbital Theory: This more advanced theory provides a more accurate description of bonding, explaining the energy levels of molecular orbitals and the distribution of electrons.
- Bond Order: The triple bond in CO signifies a strong, short bond, contributing to its high bond dissociation energy.
- Electronegativity: The difference in electronegativity between carbon and oxygen contributes to the molecule's polar character.
- Experimental Data: Spectroscopic data and other experimental measurements validate and refine our theoretical understanding of CO's structure and properties.
Conclusion: The Importance of Formal Charges in Understanding CO
The Lewis structure of carbon monoxide, with its accompanying formal charges, provides a foundational understanding of its bonding and chemical behavior. While the +1 and -1 formal charges indicate a polar molecule, it's crucial to recognize the significance of the triple bond and the substantial covalent character in the bonding. Formal charges, in conjunction with molecular orbital theory and experimental data, provide a complete picture of this seemingly simple yet remarkably complex molecule. Understanding these concepts is fundamental to interpreting its reactivity, spectroscopic properties, and its role in various chemical and biological processes. The analysis presented here serves as a valuable illustration of how simple tools like Lewis structures and formal charges can provide critical insights into the intricacies of molecular bonding and properties. The study of formal charges within the context of resonance and compared to other molecules further enhances this understanding.
Latest Posts
Latest Posts
-
How Many Electrons Are In Titanium
Apr 04, 2025
-
Two Thirds Of A Number Algebraic Expression
Apr 04, 2025
-
Is Photosynthesis A Chemical Or Physical Change
Apr 04, 2025
-
Convert 101 Degrees Fahrenheit To Celsius
Apr 04, 2025
-
How Many Minute In A Week
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Lewis Structure For Co With Formal Charges . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
