How Many Electrons Are In Titanium
listenit
Apr 04, 2025 · 5 min read
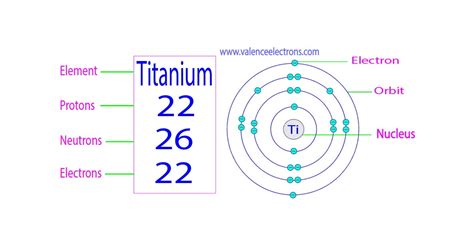
Table of Contents
How Many Electrons Are in Titanium? A Deep Dive into Atomic Structure
Titanium, a lustrous transition metal with a silvery-white color, is renowned for its strength, lightweight nature, and corrosion resistance. Understanding its properties requires delving into its atomic structure, specifically the number of electrons it possesses. This article will explore this question in detail, examining the concept of atomic number, electron configuration, and the implications of titanium's electron arrangement on its chemical behavior and applications.
Understanding Atomic Number and Electron Count
The number of electrons in an atom is directly related to its atomic number. The atomic number represents the number of protons found in the nucleus of an atom. In a neutral atom (an atom with no net electrical charge), the number of protons is equal to the number of electrons. This fundamental principle governs the chemical properties and behavior of every element.
For titanium, the atomic number is 22. This crucial piece of information immediately tells us that a neutral titanium atom contains 22 electrons.
Electron Configuration: Unveiling Titanium's Electron Arrangement
Knowing the number of electrons is only half the story. To understand titanium's properties fully, we need to explore its electron configuration. This describes how the electrons are distributed among the different energy levels and orbitals within the atom. Electrons occupy these orbitals following specific rules governed by quantum mechanics.
The electron configuration of titanium is 1s²2s²2p⁶3s²3p⁶4s²3d². Let's break this down:
- 1s²: Two electrons occupy the first energy level (n=1) in the 's' orbital. The 's' orbital can hold a maximum of two electrons.
- 2s²: Two electrons occupy the second energy level (n=2) in the 's' orbital.
- 2p⁶: Six electrons occupy the second energy level (n=2) in the three 'p' orbitals. Each 'p' orbital can hold two electrons, and there are three 'p' orbitals.
- 3s²: Two electrons occupy the third energy level (n=3) in the 's' orbital.
- 3p⁶: Six electrons occupy the third energy level (n=3) in the three 'p' orbitals.
- 4s²: Two electrons occupy the fourth energy level (n=4) in the 's' orbital.
- 3d²: Two electrons occupy the third energy level (n=3) in the five 'd' orbitals.
This electron configuration explains titanium's position in the periodic table as a transition metal. Transition metals are characterized by partially filled 'd' orbitals, which is precisely the case for titanium with its two electrons in the 3d orbitals.
Implications of Titanium's Electron Configuration on its Properties
The distribution of electrons in titanium's orbitals directly influences its chemical and physical properties:
-
Metallic Bonding: The relatively loosely held valence electrons in the 4s and 3d orbitals contribute to the metallic bonding in titanium. This type of bonding is responsible for its high strength, malleability, and ductility. The electrons are delocalized, meaning they are not bound to specific atoms but are free to move throughout the metal lattice. This allows for easy deformation and electrical conductivity.
-
Reactivity and Oxidation States: Titanium's electron configuration allows it to exhibit multiple oxidation states. The most common oxidation state is +4, where titanium loses all four of its valence electrons (two from the 4s and two from the 3d orbitals). However, titanium can also exist in +3 and +2 oxidation states, reflecting its ability to lose different numbers of electrons. This versatility contributes to its diverse chemical reactivity.
-
Corrosion Resistance: Titanium's remarkable resistance to corrosion arises from the formation of a thin, tenacious oxide layer (TiO₂) on its surface when exposed to air. This passive oxide layer acts as a barrier, preventing further oxidation and protecting the underlying metal from degradation. This exceptional corrosion resistance makes titanium ideal for applications in harsh environments.
Titanium in Different Applications: A Showcase of its Properties
The unique combination of strength, lightweight nature, and corrosion resistance makes titanium highly sought-after in a wide range of applications:
-
Aerospace Industry: Titanium alloys are extensively used in aircraft and spacecraft components due to their high strength-to-weight ratio. This allows for lighter and more fuel-efficient vehicles.
-
Medical Implants: Titanium's biocompatibility and corrosion resistance make it a preferred material for medical implants, such as artificial joints, dental implants, and surgical instruments. Its inertness minimizes the risk of rejection or adverse reactions in the body.
-
Chemical Processing: The excellent corrosion resistance of titanium makes it suitable for handling corrosive chemicals and handling processes in various industries.
-
Sporting Goods: Titanium's lightweight strength is used to create high-performance sporting goods like golf clubs, bicycle frames, and tennis rackets.
-
Jewelry: Its attractive silvery-white color, resistance to tarnish, and hypoallergenic nature have led to its use in high-end jewelry.
Isotopes and Electron Count Variations
While a neutral titanium atom has 22 electrons, it's important to note that titanium exists in several isotopic forms. Isotopes are atoms of the same element that have the same number of protons but differ in the number of neutrons. The number of neutrons affects the atom's mass but not its electron configuration or chemical properties in a neutral state. Therefore, even though titanium has several isotopes (e.g., ⁴⁶Ti, ⁴⁷Ti, ⁴⁸Ti, ⁴⁹Ti, and ⁵⁰Ti), a neutral atom of any of these isotopes will still possess 22 electrons.
However, if titanium becomes an ion (a charged atom), the electron count changes. For example, a Ti⁴⁺ ion (titanium with a +4 charge) has lost four electrons and thus has only 18 electrons.
Conclusion: The Significance of 22 Electrons in Titanium
The fact that a neutral titanium atom possesses 22 electrons is paramount to understanding its chemical and physical properties. This electron count, along with its electron configuration, dictates its metallic bonding, reactivity, oxidation states, and ultimately, its diverse applications across various industries. From aerospace to medicine, the unique characteristics stemming from this precise number of electrons have established titanium as a vital material in modern technology. This deep dive highlights the importance of atomic structure and electron configuration in explaining the behavior and utility of elements like titanium. The seemingly simple number, 22, holds the key to understanding the extraordinary properties of this remarkable metal. Further research into titanium and other transition metals will continue to unveil new possibilities and applications, solidifying its importance in shaping technological advancements.
Latest Posts
Latest Posts
-
Least Common Multiple Of 7 2 And 3
Apr 04, 2025
-
What Is The Purpose Of The Experiment
Apr 04, 2025
-
Why Must The Remainder Be Less Than The Divisor
Apr 04, 2025
-
How Many Inches In Two Yards
Apr 04, 2025
-
What Is 9 10 In Decimal Form
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about How Many Electrons Are In Titanium . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
