Is Photosynthesis A Chemical Or Physical Change
listenit
Apr 04, 2025 · 5 min read
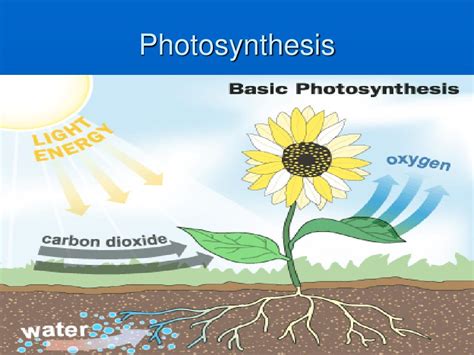
Table of Contents
Is Photosynthesis a Chemical or Physical Change? A Deep Dive
Photosynthesis, the remarkable process by which green plants and some other organisms use sunlight to synthesize foods from carbon dioxide and water, is a cornerstone of life on Earth. But is this vital process a chemical change or a physical change? The answer, as we'll explore in detail, is unequivocally chemical. While physical changes alter the form or appearance of a substance without changing its chemical composition, chemical changes result in the formation of entirely new substances with different properties. Photosynthesis perfectly exemplifies a chemical change due to the fundamental transformation of reactants into entirely new products.
Understanding Chemical and Physical Changes
Before delving into the specifics of photosynthesis, let's establish a clear understanding of the distinction between chemical and physical changes.
Physical Changes: A Change in Form, Not Substance
Physical changes involve alterations in the physical properties of a substance, such as its shape, size, state (solid, liquid, gas), or texture. These changes are generally reversible. Examples include:
- Melting ice: Ice (solid water) transforms into liquid water, but the chemical composition remains H₂O.
- Boiling water: Liquid water changes to water vapor (steam), again without altering its chemical makeup.
- Crushing a can: The shape of the can changes, but the metal remains the same.
- Dissolving sugar in water: The sugar disappears into the water, but its chemical structure is unchanged; it can be recovered by evaporation.
Chemical Changes: New Substances are Formed
Chemical changes, also known as chemical reactions, involve the rearrangement of atoms and molecules to form entirely new substances with different properties. These changes are often irreversible. Evidence of a chemical change includes:
- Formation of a gas: The production of bubbles or a noticeable odor.
- Formation of a precipitate: The formation of a solid from a solution.
- Color change: A significant and permanent alteration in color.
- Temperature change: A noticeable increase (exothermic reaction) or decrease (endothermic reaction) in temperature.
- Change in odor: A new and distinct smell.
Photosynthesis: A Detailed Look at a Chemical Transformation
Photosynthesis, the process by which green plants and certain other organisms convert light energy into chemical energy in the form of glucose (a sugar), undeniably fits the criteria for a chemical change. Let's examine why:
The Reactants and Products: A Clear Indication of Chemical Change
Photosynthesis begins with simple inorganic reactants:
- Carbon dioxide (CO₂): A gas absorbed from the atmosphere.
- Water (H₂O): Absorbed through the roots from the soil.
In the presence of sunlight and chlorophyll (the green pigment in plants), these reactants undergo a series of complex reactions. The end products are:
- Glucose (C₆H₁₂O₆): A complex organic molecule, a sugar that serves as the plant's primary energy source.
- Oxygen (O₂): A gas released into the atmosphere.
The transformation from simple inorganic molecules (CO₂ and H₂O) to complex organic molecules (C₆H₁₂O₆) and a completely different gas (O₂) is a definitive hallmark of a chemical change. The atoms from the reactants are rearranged to form entirely new molecules with different chemical properties. Glucose has vastly different properties than carbon dioxide and water; it's a source of energy, while the original reactants are not. Oxygen, similarly, plays a completely different biological role than water.
Energy Transformation: Another Key to Understanding Chemical Changes
Photosynthesis involves a crucial energy transformation. Light energy from the sun is absorbed by chlorophyll and converted into chemical energy stored in the bonds of the glucose molecule. This energy conversion is a characteristic feature of chemical reactions. Physical changes, on the other hand, do not involve significant energy transformations of this nature.
Irreversibility: A Further Indication of Chemical Change
While some physical changes can be reversed (e.g., melting and freezing water), the process of photosynthesis is not easily reversed in its entirety. While glucose can be broken down through cellular respiration (another chemical process), recreating the precise conditions and steps of photosynthesis to directly reform carbon dioxide and water from glucose and oxygen is not a simple reversal.
The Role of Enzymes: Catalysts in Chemical Reactions
Photosynthesis involves a complex series of reactions catalyzed by enzymes. Enzymes are biological catalysts that speed up chemical reactions without being consumed themselves. The presence of enzymes further solidifies the understanding that photosynthesis is a chemical process. Physical changes generally do not involve enzymatic catalysts.
Addressing Potential Misconceptions
Some might argue that the physical process of light absorption by chlorophyll plays a role. While light absorption is a physical phenomenon, it initiates the chemical reactions that define photosynthesis. The absorption of light energy is a necessary trigger for the subsequent chemical changes, but it is not the defining characteristic of the overall process.
Another potential point of confusion might be the involvement of water in photosynthesis. Water is a reactant, undergoing a chemical transformation. Its role in the process isn't simply a matter of a physical change like dissolving a substance; it's a fundamental part of the chemical reactions that create glucose and oxygen.
Conclusion: Photosynthesis is Undeniably a Chemical Change
In conclusion, there's no ambiguity: photosynthesis is a chemical change. The process involves the transformation of simple inorganic reactants (carbon dioxide and water) into complex organic products (glucose and oxygen) through a series of enzyme-catalyzed reactions driven by light energy. This transformation produces entirely new substances with different properties, making it a clear-cut example of a chemical reaction. Understanding this fundamental distinction is crucial for comprehending the intricate processes that sustain life on our planet. The chemical nature of photosynthesis highlights its profound impact on the Earth's ecosystems and the flow of energy throughout the biosphere. The production of oxygen, a byproduct of this vital chemical process, has shaped the atmosphere and made possible the evolution of oxygen-dependent life forms. Therefore, appreciating photosynthesis as a fundamental chemical process unlocks a deeper understanding of its significance in shaping our world.
Latest Posts
Latest Posts
-
How Do You Find Ml Quantum Number
Apr 04, 2025
-
How Many Unpaired Electrons Does Manganese Have
Apr 04, 2025
-
Water Is Made Up Of Which Two Elements
Apr 04, 2025
-
What Is 9 Percent Of 50
Apr 04, 2025
-
Distance From Earth To Pluto In Light Years
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Is Photosynthesis A Chemical Or Physical Change . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
