Is The Limiting Reactant The One With Less Moles
listenit
Apr 05, 2025 · 5 min read
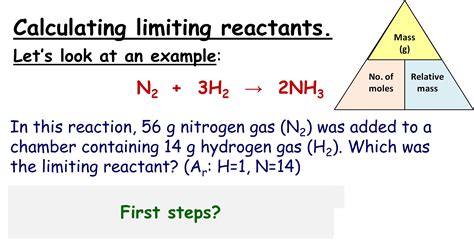
Table of Contents
Is the Limiting Reactant the One with Less Moles? A Deep Dive into Stoichiometry
The question, "Is the limiting reactant the one with fewer moles?" is a common misconception in stoichiometry. While the reactant with fewer moles might be the limiting reactant, it's not always the case. This article will delve deep into the concept of limiting reactants, explaining why simply comparing mole amounts is insufficient and outlining the correct method to identify the limiting reactant in any chemical reaction.
Understanding Limiting Reactants
In a chemical reaction, reactants are the substances consumed during the reaction to produce products. A stoichiometric reaction occurs when all reactants are completely consumed, leaving no excess. However, in reality, chemical reactions often involve an excess of one or more reactants. The reactant that is completely consumed first and limits the amount of product formed is called the limiting reactant. The other reactants are present in excess.
The limiting reactant dictates the theoretical yield, which is the maximum amount of product that can be formed based on the stoichiometry of the balanced chemical equation. Any reactant in excess will be left unreacted after the reaction is complete.
Why Comparing Moles Alone is Insufficient
While it's tempting to assume the reactant with fewer moles is the limiting reactant, this is a flawed approach. The stoichiometric coefficients in a balanced chemical equation determine the ratio in which reactants combine. Consider this example:
Reaction: 2H₂ + O₂ → 2H₂O
Let's say we have 2 moles of H₂ and 2 moles of O₂. Comparing moles alone would suggest H₂ is the limiting reactant because it has the same number of moles as O₂, but this is incorrect.
According to the balanced equation, 2 moles of H₂ react with 1 mole of O₂. Therefore, 2 moles of H₂ require only 1 mole of O₂. Since we have 2 moles of O₂, O₂ is in excess, and H₂ is the limiting reactant.
The crucial factor is the mole ratio defined by the coefficients in the balanced equation. We must compare the moles of each reactant to the stoichiometric ratio to determine the limiting reactant accurately.
Determining the Limiting Reactant: A Step-by-Step Approach
Here's a systematic approach to identifying the limiting reactant:
-
Balance the Chemical Equation: Ensure the chemical equation is correctly balanced. This is critical as the coefficients determine the mole ratios.
-
Convert Quantities to Moles: If the given quantities of reactants are not in moles, convert them to moles using their molar masses.
-
Determine the Mole Ratio: From the balanced equation, determine the mole ratio between the reactants.
-
Compare Mole Ratios: For each reactant, divide the number of moles present by its stoichiometric coefficient from the balanced equation.
-
Identify the Limiting Reactant: The reactant with the smallest calculated ratio from step 4 is the limiting reactant. This reactant will be completely consumed first, limiting the amount of product formed.
Illustrative Examples
Let's work through a couple of examples to solidify the concept:
Example 1:
Reaction: N₂ + 3H₂ → 2NH₃
We have 5 moles of N₂ and 10 moles of H₂.
- Step 1: The equation is already balanced.
- Step 2: Quantities are already in moles.
- Step 3: The mole ratio of N₂ to H₂ is 1:3.
- Step 4: For N₂: 5 moles / 1 = 5. For H₂: 10 moles / 3 ≈ 3.33.
- Step 5: Since 3.33 < 5, H₂ is the limiting reactant.
Example 2:
Reaction: 2Fe + 3Cl₂ → 2FeCl₃
We have 10 grams of Fe and 10 grams of Cl₂. Molar mass of Fe is approximately 56 g/mol, and the molar mass of Cl₂ is approximately 71 g/mol.
- Step 1: The equation is already balanced.
- Step 2: Convert grams to moles: Moles of Fe = 10 g / 56 g/mol ≈ 0.18 moles. Moles of Cl₂ = 10 g / 71 g/mol ≈ 0.14 moles.
- Step 3: The mole ratio of Fe to Cl₂ is 2:3.
- Step 4: For Fe: 0.18 moles / 2 = 0.09. For Cl₂: 0.14 moles / 3 ≈ 0.05.
- Step 5: Since 0.05 < 0.09, Cl₂ is the limiting reactant.
Beyond Simple Reactions: More Complex Scenarios
The principles remain the same even with more complex reactions involving multiple reactants. Always follow the step-by-step process, ensuring you carefully consider the stoichiometric coefficients for each reactant.
For reactions with more than two reactants, you will repeat the steps above for each reactant and compare the calculated ratios to identify the limiting reactant.
Practical Applications and Real-World Significance
Understanding limiting reactants is crucial in various fields:
-
Industrial Chemistry: Optimizing chemical processes to maximize product yield and minimize waste requires accurate determination of the limiting reactant. This impacts efficiency, cost, and environmental impact.
-
Pharmaceutical Industry: Precise stoichiometry is vital in drug synthesis to ensure the desired product is obtained in the correct amount and purity. Identifying the limiting reactant is crucial for controlling reaction efficiency and quality.
-
Environmental Science: Understanding limiting reactants helps in modeling environmental processes, such as pollutant degradation or nutrient cycling. Knowing the limiting factor can inform strategies for remediation or resource management.
-
Agricultural Science: In fertilizers, the limiting nutrient often determines plant growth. Identifying the limiting nutrient helps optimize fertilizer application for maximum crop yield.
Conclusion
In conclusion, simply comparing the number of moles of reactants is not sufficient to determine the limiting reactant. The correct method involves careful consideration of the balanced chemical equation, mole ratios, and a systematic comparison of the moles of each reactant relative to their stoichiometric coefficients. Mastering this concept is crucial for accurately predicting reaction yields, optimizing processes, and understanding various chemical and environmental phenomena. This understanding is vital in many scientific and industrial applications, highlighting the practical significance of stoichiometry in our world.
Latest Posts
Latest Posts
-
How To Tell If A Molecule Is Planar
Apr 06, 2025
-
Which Kingdom S Include Organisms That Are Autotrophic Or Heterotrophic
Apr 06, 2025
-
Odds Of Rolling 5 Dice The Same
Apr 06, 2025
-
Area Of Circle With Radius Of 8
Apr 06, 2025
-
Compare And Contrast Synthesis And Decomposition Reactions
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about Is The Limiting Reactant The One With Less Moles . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
