How To Tell If A Molecule Is Planar
listenit
Apr 06, 2025 · 5 min read
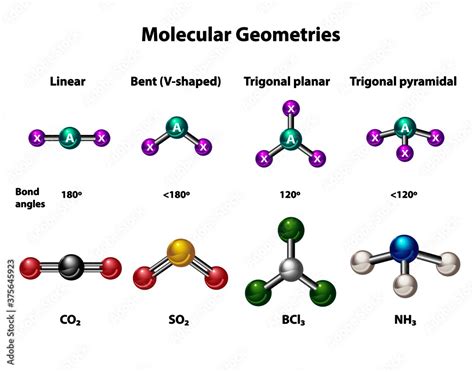
Table of Contents
How to Tell if a Molecule is Planar: A Comprehensive Guide
Determining whether a molecule is planar or not is a crucial concept in chemistry, impacting its properties and reactivity. Planarity, or the tendency for atoms in a molecule to lie in the same plane, influences factors like its dipole moment, its ability to undergo certain reactions, and its spectral properties (UV-Vis, NMR). This guide provides a comprehensive approach to determining molecular planarity, encompassing various techniques and considerations.
Understanding Molecular Geometry and Planarity
Before delving into the specifics, it's vital to understand the fundamental principles governing molecular geometry. Molecular geometry describes the three-dimensional arrangement of atoms in a molecule. This arrangement is primarily dictated by valence shell electron pair repulsion (VSEPR) theory, which postulates that electron pairs (both bonding and lone pairs) around a central atom will arrange themselves to minimize electrostatic repulsion, thus maximizing the distance between them.
A molecule is considered planar if all its constituent atoms lie within a single plane. This doesn't necessarily mean that the molecule is perfectly flat; slight deviations may occur due to thermal vibrations or other subtle factors, but the overall structure retains its essential planar nature.
Key Methods to Determine Planarity
Several methods exist to determine if a molecule is planar. These methods range from simple visual inspection (for simpler molecules) to complex computational analyses (for more intricate structures):
1. Visual Inspection and VSEPR Theory
For smaller, simpler molecules, visual inspection coupled with VSEPR theory can be sufficient. Look for molecules with a central atom surrounded by atoms that exhibit specific bonding patterns.
-
Trigonal planar geometry: Molecules with a central atom bonded to three other atoms with no lone pairs exhibit trigonal planar geometry, making them planar. Examples include BF₃ (boron trifluoride) and SO₃ (sulfur trioxide). The bond angles are approximately 120°.
-
Linear geometry: Molecules with a central atom bonded to two other atoms with no lone pairs exhibit linear geometry and are planar. Examples include CO₂ (carbon dioxide) and BeCl₂ (beryllium chloride). The bond angle is 180°.
Important Note: The presence of lone pairs significantly impacts the geometry and planarity. While a molecule with a central atom surrounded by three atoms and no lone pairs is always planar, the presence of a lone pair will invariably distort the geometry from planar.
2. Hybridization and Planarity
The concept of hybridization plays a pivotal role in predicting molecular geometry and planarity. Hybridization involves the mixing of atomic orbitals to form new hybrid orbitals that are involved in bonding.
-
sp² hybridization: Atoms with sp² hybridization typically form trigonal planar geometries, resulting in planar molecules. This hybridization is common in molecules containing double bonds (C=C, C=O, etc.).
-
sp hybridization: Atoms with sp hybridization generally form linear geometries, which are also planar. This hybridization is found in molecules with triple bonds (C≡C, C≡N, etc.).
-
sp³ hybridization: Atoms with sp³ hybridization usually form tetrahedral geometries, which are generally not planar. Exceptions exist, but they are less common. This hybridization is common in alkanes and other saturated molecules.
3. Resonance Structures and Delocalization
Resonance structures play a vital role in determining the planarity of some molecules. If a molecule exhibits resonance, implying delocalized electrons, the molecule will tend to be planar. This is because delocalization requires the p-orbitals involved to be parallel, which necessitates planarity. Benzene (C₆H₆) is a classic example; its delocalized π-electrons necessitate a planar structure.
4. Molecular Orbital Theory (MOT)
Molecular orbital theory provides a more sophisticated approach to understanding molecular geometry and planarity. MOT considers the interactions of atomic orbitals to form molecular orbitals that encompass the entire molecule. The resulting molecular orbitals dictate the overall electronic structure and geometry. While conceptually more complex, MOT offers a more accurate description of the electronic structure and, by extension, the planarity of molecules.
5. Spectroscopic Techniques
Various spectroscopic techniques, such as X-ray crystallography, NMR spectroscopy, and UV-Vis spectroscopy, can provide experimental evidence of planarity.
-
X-ray crystallography: This technique determines the precise three-dimensional structure of a molecule, providing definitive proof of planarity.
-
NMR spectroscopy: NMR coupling constants can provide information about the dihedral angles between atoms; a planar molecule will exhibit specific coupling patterns.
-
UV-Vis spectroscopy: The absorption of UV-Vis light is often influenced by the degree of conjugation and planarity. Planar conjugated molecules typically exhibit characteristic absorption bands.
Examples Illustrating Planarity
Let's analyze some examples to reinforce the understanding of planarity:
Planar Molecules:
- Benzene (C₆H₆): The delocalized π-electrons necessitate a planar structure.
- Formaldehyde (CH₂O): The sp² hybridized carbon atom forms a trigonal planar geometry.
- Ethene (C₂H₄): The sp² hybridized carbon atoms and the double bond contribute to its planarity.
- Carbon dioxide (CO₂): The linear arrangement of atoms makes it planar.
Non-Planar Molecules:
- Methane (CH₄): The tetrahedral arrangement of atoms around the central carbon atom makes it non-planar.
- Ammonia (NH₃): The presence of a lone pair on the nitrogen atom distorts the tetrahedral geometry, making it non-planar (though close to planar).
- Water (H₂O): Similar to ammonia, the lone pairs on the oxygen atom significantly distort the tetrahedral arrangement, resulting in a bent and non-planar structure.
Exceptions and Considerations
While the methods outlined above offer a robust framework for determining planarity, it's essential to acknowledge exceptions and nuances:
-
Steric hindrance: In larger molecules, steric hindrance (repulsion between atoms or groups) can slightly distort the ideal geometry, causing deviations from perfect planarity.
-
Conformational isomers: Some molecules can exist in multiple conformations, some planar and some non-planar. The predominant conformation under specific conditions determines the overall planarity.
Conclusion
Determining whether a molecule is planar involves a combination of theoretical understanding and practical application. While VSEPR theory and hybridization provide initial predictions, more sophisticated techniques like molecular orbital theory and experimental methods like X-ray crystallography often become necessary for accurate determination, especially for larger and more complex molecules. A thorough understanding of these methods allows chemists to predict and interpret the properties and reactivity of molecules, impacting diverse fields from drug discovery to materials science. Remember that the presence of lone pairs often significantly alters the geometry and planarity, and the concept of resonance plays a critical role in some instances. By employing a systematic approach incorporating multiple techniques, one can confidently assess the planarity of a given molecule.
Latest Posts
Latest Posts
-
What Is The Decimal Of 8 25
Apr 06, 2025
-
Two 20 0 G Ice Cubes At
Apr 06, 2025
-
How Much Is 1000 Days In Years
Apr 06, 2025
-
What Is 12 5 As A Decimal
Apr 06, 2025
-
8 More Than The Product Of 2 And X
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about How To Tell If A Molecule Is Planar . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
