Compare And Contrast Synthesis And Decomposition Reactions.
listenit
Apr 06, 2025 · 5 min read
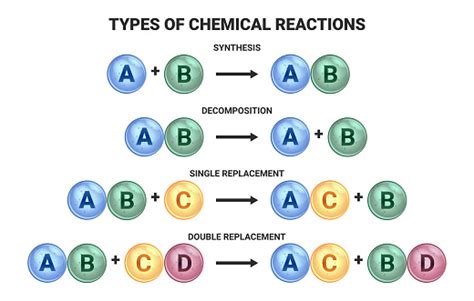
Table of Contents
Compare and Contrast Synthesis and Decomposition Reactions
Chemical reactions are the fundamental processes that govern the transformation of matter. Understanding these reactions is crucial in various fields, from medicine and materials science to environmental science and engineering. Among the many types of chemical reactions, synthesis and decomposition reactions represent two fundamental and contrasting processes. This article will delve into a detailed comparison and contrast of these reactions, exploring their mechanisms, applications, and significance in chemistry.
What is a Synthesis Reaction?
A synthesis reaction, also known as a combination reaction, is a type of chemical reaction where two or more simple substances combine to form a more complex product. The general form of a synthesis reaction can be represented as:
A + B → AB
Where A and B are reactants and AB is the product. This simple equation encapsulates the core principle: the reactants combine to create a single, more complex product. Importantly, the product possesses different chemical and physical properties than the reactants.
Characteristics of Synthesis Reactions:
- Combination of reactants: The defining feature is the combination of two or more substances. These substances can be elements or simpler compounds.
- Formation of a single product: Only one product is formed as a result of the reaction.
- Energy changes: Synthesis reactions can be either exothermic (releasing heat) or endothermic (absorbing heat). The energy change depends on the specific reactants and the strength of the bonds formed in the product.
- Often irreversible: While some synthesis reactions can be reversed under specific conditions, many are effectively irreversible under normal circumstances.
Examples of Synthesis Reactions:
-
Formation of water: The reaction between hydrogen gas and oxygen gas to form water is a classic example:
2H₂ + O₂ → 2H₂O
-
Formation of metal oxides: Metals readily react with oxygen to form metal oxides. For instance, the reaction of iron with oxygen:
4Fe + 3O₂ → 2Fe₂O₃
-
Formation of salts: The reaction between a metal and a non-metal often results in the formation of a salt. For example, the reaction of sodium and chlorine:
2Na + Cl₂ → 2NaCl
-
Formation of ammonia: The Haber-Bosch process, a crucial industrial process for producing ammonia fertilizer, involves a synthesis reaction:
N₂ + 3H₂ → 2NH₃
What is a Decomposition Reaction?
In contrast to synthesis reactions, a decomposition reaction involves the breakdown of a single, more complex compound into two or more simpler substances. This process often requires an input of energy, such as heat, light, or electricity. The general form of a decomposition reaction is:
AB → A + B
Where AB is the reactant and A and B are the products. This highlights the reverse nature of decomposition compared to synthesis.
Characteristics of Decomposition Reactions:
- Breakdown of a single reactant: The key characteristic is the breakdown of one compound into two or more simpler substances.
- Formation of multiple products: Unlike synthesis, decomposition always results in multiple products.
- Energy requirement: Decomposition reactions typically require an external energy source to initiate and sustain the reaction. This energy overcomes the bond strength within the reactant molecule.
- Often reversible: While not always the case, some decomposition reactions can be reversed under specific conditions, leading back to the original compound.
Examples of Decomposition Reactions:
-
Electrolysis of water: Passing an electric current through water breaks it down into hydrogen and oxygen gases:
2H₂O → 2H₂ + O₂
-
Thermal decomposition of carbonates: Heating metal carbonates, such as calcium carbonate, leads to their decomposition:
CaCO₃ → CaO + CO₂
-
Decomposition of hydrogen peroxide: Hydrogen peroxide spontaneously decomposes into water and oxygen, though this process can be accelerated by catalysts:
2H₂O₂ → 2H₂O + O₂
-
Decomposition of potassium chlorate: Heating potassium chlorate produces potassium chloride and oxygen gas:
2KClO₃ → 2KCl + 3O₂
Comparing Synthesis and Decomposition Reactions: A Detailed Overview
| Feature | Synthesis Reaction | Decomposition Reaction |
|---|---|---|
| Type of Reaction | Combination | Breakdown |
| Number of Reactants | Two or more | One |
| Number of Products | One | Two or more |
| Energy Change | Can be exothermic or endothermic | Typically endothermic (requires energy input) |
| Reversibility | Often irreversible, but some are reversible | Often reversible, but some are irreversible |
| General Equation | A + B → AB | AB → A + B |
| Examples | Formation of water, metal oxides, salts, ammonia | Electrolysis of water, thermal decomposition of carbonates, decomposition of hydrogen peroxide |
Contrasting Synthesis and Decomposition: Key Differences
While both are fundamental reaction types, several key differences distinguish synthesis and decomposition reactions:
-
Directionality: Synthesis reactions build up complex molecules from simpler ones, while decomposition reactions break down complex molecules into simpler ones. This represents a fundamental difference in the direction of the chemical change.
-
Energy requirements: Synthesis reactions can be either exothermic or endothermic, while decomposition reactions are usually endothermic, requiring energy to break the bonds in the reactant molecule. This energy input is a crucial distinguishing factor.
-
Number of products and reactants: Synthesis reactions always have more reactants than products, while decomposition reactions always have more products than reactants. This simple count provides a quick method for identifying the reaction type.
Applications of Synthesis and Decomposition Reactions
Both synthesis and decomposition reactions have widespread applications across numerous fields:
Synthesis Reactions:
- Industrial chemistry: The production of ammonia (Haber-Bosch process), sulfuric acid, and various polymers relies heavily on synthesis reactions.
- Materials science: The creation of new materials with specific properties often involves carefully controlled synthesis reactions.
- Pharmaceutical industry: The synthesis of drugs and medicines involves complex multi-step synthesis reactions.
- Metallurgy: The production of alloys and other metallic materials uses synthesis reactions.
Decomposition Reactions:
- Extraction of metals: The extraction of metals from their ores often involves decomposition reactions, such as the reduction of metal oxides.
- Production of oxygen: The decomposition of potassium chlorate is a common method for producing oxygen in laboratories.
- Analysis of compounds: Decomposition reactions are used in analytical chemistry to identify the components of unknown compounds.
- Environmental remediation: Certain decomposition reactions are used to break down pollutants and other harmful substances.
Conclusion: Synthesis and Decomposition – Two Sides of the Same Coin
Synthesis and decomposition reactions represent two fundamental and opposing types of chemical transformations. While synthesis reactions construct complex molecules from simpler ones, decomposition reactions break down complex molecules into simpler components. Understanding these reactions and their characteristics is essential for comprehending chemical processes in various contexts, from industrial manufacturing to biological systems. Their contrasting natures highlight the dynamic equilibrium and intricate interplay of chemical reactions that shape our world. The energy considerations, reversibility aspects, and diverse applications of both reaction types underscore their fundamental importance in chemistry and related fields.
Latest Posts
Latest Posts
-
What Is The Decimal Of 8 25
Apr 06, 2025
-
Two 20 0 G Ice Cubes At
Apr 06, 2025
-
How Much Is 1000 Days In Years
Apr 06, 2025
-
What Is 12 5 As A Decimal
Apr 06, 2025
-
8 More Than The Product Of 2 And X
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about Compare And Contrast Synthesis And Decomposition Reactions. . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
