Is Koh A Base Or Acid
listenit
Apr 02, 2025 · 5 min read
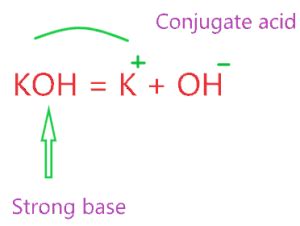
Table of Contents
Is KOH a Base or Acid? Understanding Strong Bases and Their Properties
Potassium hydroxide (KOH), also known as caustic potash, is a highly alkaline substance. But what does that really mean? Let's delve deep into the world of acids and bases to definitively answer the question: Is KOH a base or an acid? And beyond that, let's explore its properties, applications, and safety precautions.
Understanding Acids and Bases
Before we label KOH, it's crucial to understand the fundamental concepts of acids and bases. Several theories define these, but the most relevant for our purpose are the Arrhenius and Brønsted-Lowry theories.
Arrhenius Theory
The Arrhenius theory defines acids as substances that produce hydrogen ions (H⁺) when dissolved in water, and bases as substances that produce hydroxide ions (OH⁻) when dissolved in water. This is a relatively simple definition, but it lays the groundwork for understanding acid-base reactions.
Brønsted-Lowry Theory
The Brønsted-Lowry theory offers a broader perspective. It defines acids as proton (H⁺) donors and bases as proton acceptors. This theory expands the definition beyond just aqueous solutions, encompassing a wider range of reactions.
KOH and the Definitions
According to both the Arrhenius and Brønsted-Lowry theories, KOH is unequivocally a base. When dissolved in water, KOH dissociates completely into potassium ions (K⁺) and hydroxide ions (OH⁻). This abundance of hydroxide ions makes it a strong base.
The Strength of KOH: A Strong Base
The term "strong base" signifies complete dissociation in water. KOH is a classic example. It doesn't just partially break apart into ions; it almost entirely separates, maximizing the concentration of OH⁻ ions in the solution. This characteristic directly influences its reactivity and pH.
pH and pOH: Measuring Alkalinity
The pH scale measures the acidity or alkalinity of a solution. It ranges from 0 to 14, with 7 being neutral. Values below 7 indicate acidity, and values above 7 indicate alkalinity. The pOH scale is complementary, representing the hydroxide ion concentration. A high pOH value corresponds to a low hydroxide ion concentration (more acidic), and vice versa. A strong base like KOH will have a high pH value and a low pOH value.
The Reaction of KOH with Water
The dissociation of KOH in water is a highly exothermic reaction, meaning it releases heat. This is a characteristic of strong bases, reflecting their energetic interaction with water molecules. The reaction is simple and complete:
KOH(s) → K⁺(aq) + OH⁻(aq)
This equation shows the solid KOH (s) dissolving in water (aq) to produce potassium cations (K⁺) and hydroxide anions (OH⁻).
Applications of Potassium Hydroxide
KOH's strong basic nature makes it incredibly versatile with applications in various fields:
1. Industrial Applications
- Soap and Detergent Production: KOH is a crucial ingredient in the saponification process, converting fats and oils into soap. Its strong basicity facilitates the reaction, creating the characteristic soap molecules.
- Fertilizer Production: In agriculture, KOH contributes to the production of potassium-based fertilizers, providing an essential nutrient for plant growth.
- Food Processing: Although seemingly counterintuitive, KOH finds use in food processing, such as modifying the texture of food items or controlling pH in certain products.
- Chemical Synthesis: The strong base nature is fundamental in many chemical reactions and syntheses, acting as a catalyst or reactant.
2. Laboratory Applications
- Titrations: KOH solutions are frequently used in titrations to determine the concentration of acids. Its complete dissociation ensures precise measurements.
- pH Adjustments: In laboratories, it helps maintain a specific pH range in reactions or experiments, demonstrating its importance in pH control.
- Synthesis of Organic Compounds: KOH's role in organic chemistry synthesis is significant, facilitating numerous reactions involving the formation of carbon-carbon bonds and other crucial transformations.
3. Other Applications
- Battery Manufacturing: KOH plays a critical role in alkaline batteries, acting as an electrolyte, facilitating the flow of ions and maintaining the electrical current.
- Electroplating: In electroplating processes, KOH is sometimes utilized for the cleaning and preparation of metal surfaces before coating.
Safety Precautions when Handling KOH
KOH is a corrosive substance and requires careful handling. Always follow these safety guidelines:
- Wear appropriate personal protective equipment (PPE): This includes safety goggles, gloves (chemical-resistant), and a lab coat to prevent skin and eye contact.
- Work in a well-ventilated area: KOH solutions can release irritating vapors. Proper ventilation is crucial to minimize inhalation hazards.
- Handle with care: Avoid direct contact with skin, eyes, or clothing. Spills should be cleaned immediately with ample water, and appropriate neutralizing agents might be required.
- Store properly: Store KOH in a tightly sealed container in a cool, dry place, away from incompatible materials.
Understanding the Implications of KOH's Strong Basicity
The strong basic nature of KOH has profound consequences for its reactivity and applications:
- Reaction with Acids: KOH readily reacts with acids in neutralization reactions, producing salt and water. This reaction is highly exothermic, releasing considerable heat.
- Reaction with Metals: KOH can react with certain metals, especially amphoteric metals (like aluminum and zinc), producing hydrogen gas and metal hydroxides. This reaction can be dangerous due to the flammability of hydrogen gas.
- Saponification Reaction: The reaction with fats and oils in saponification produces soap, demonstrating its role in everyday products.
- Corrosion: KOH's corrosive nature mandates careful handling to avoid damage to materials.
Distinguishing KOH from Other Bases
While KOH is a strong base, it’s important to differentiate it from other bases:
- NaOH (Sodium Hydroxide): Both KOH and NaOH are strong bases, but they have slightly different properties impacting their applications. NaOH is often preferred in some industrial processes.
- Ammonia (NH₃): Ammonia is a weak base, unlike the strong basicity of KOH. It does not dissociate completely in water.
- Calcium Hydroxide (Ca(OH)₂): While also a base, Ca(OH)₂ is less soluble in water than KOH, making its hydroxide ion concentration lower.
Conclusion: KOH – A Powerful and Versatile Base
In conclusion, KOH is undeniably a strong base. Its complete dissociation in water, producing a high concentration of hydroxide ions, defines its strong alkalinity. This property underpins its diverse applications across numerous industries and laboratories. However, its corrosive nature demands rigorous safety precautions to mitigate potential hazards. Understanding its properties and handling procedures is crucial for its safe and effective utilization. Remember to always prioritize safety when working with KOH or any other strong base. The knowledge gained from understanding its chemical nature allows for its responsible and beneficial application.
Latest Posts
Latest Posts
-
Which State Of Matter Takes The Shape Of Its Container
Apr 03, 2025
-
Whats The Square Root Of 145
Apr 03, 2025
-
What Is The Highest Common Factor Of 24 And 32
Apr 03, 2025
-
X 3y 9 In Slope Intercept Form
Apr 03, 2025
-
What State Of Matter Is Electricity
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Is Koh A Base Or Acid . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
