Identify The Characteristics Of The Hydroboration-oxidation Of An Alkene
listenit
Apr 05, 2025 · 5 min read
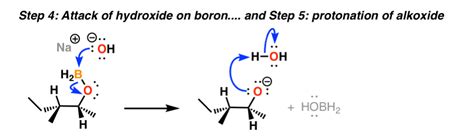
Table of Contents
Identifying the Characteristics of the Hydroboration-Oxidation of an Alkene
The hydroboration-oxidation of alkenes is a powerful and versatile reaction in organic chemistry, renowned for its ability to perform anti-Markovnikov addition of water across a carbon-carbon double bond. Understanding its characteristics is crucial for predicting reaction outcomes and designing synthetic pathways. This comprehensive guide delves into the intricacies of this reaction, exploring its mechanism, regioselectivity, stereoselectivity, and limitations.
Understanding the Reaction Mechanism: A Step-by-Step Approach
The hydroboration-oxidation reaction proceeds in two distinct steps: hydroboration and oxidation. Let's examine each stage in detail:
Step 1: Hydroboration
This step involves the addition of borane (BH₃) or a borane derivative, such as 9-borabicyclo[3.3.1]nonane (9-BBN) or disiamylborane, to the alkene. Borane, a Lewis acid, readily reacts with the alkene's electron-rich pi bond. The reaction is a concerted process, meaning it occurs in a single step without the formation of intermediates.
-
Concerted Mechanism: The boron atom attacks one carbon atom of the double bond, while a hydride (H⁻) from the borane simultaneously attacks the other carbon atom. This simultaneous addition is crucial for the reaction's stereochemistry.
-
Syn Addition: Both the boron and the hydrogen add to the same face of the alkene, resulting in syn addition. This is a hallmark characteristic differentiating hydroboration-oxidation from other alkene hydration methods.
-
Regioselectivity: The addition follows an anti-Markovnikov regioselectivity. This means the hydrogen atom adds to the more substituted carbon atom of the double bond, while the boron atom adds to the less substituted carbon. This is contrary to Markovnikov's rule, which predicts the addition of the hydrogen to the less substituted carbon.
-
Steric Hindrance: The steric bulk of the borane reagent influences the regioselectivity. Bulky boranes, such as 9-BBN, exhibit enhanced regioselectivity, preferentially adding to the less hindered side of the alkene.
Step 2: Oxidation
The organoborane intermediate formed in the hydroboration step is then oxidized using an oxidizing agent, typically alkaline hydrogen peroxide (H₂O₂). This step replaces the boron atom with a hydroxyl group (-OH).
-
Mechanism of Oxidation: The oxidation involves a series of steps that ultimately lead to the replacement of the boron atom with a hydroxyl group. The exact mechanism can be complex and dependent on the oxidizing agent employed, but it generally involves the formation of a boronate ester followed by its hydrolysis.
-
Stereochemistry Preservation: The oxidation step generally proceeds without affecting the stereochemistry established in the hydroboration step. The syn addition of boron and hydrogen in the first step is preserved, leading to a specific stereochemical outcome in the final alcohol product.
Regioselectivity and Stereoselectivity: Key Characteristics
The hydroboration-oxidation reaction is highly regioselective and stereoselective. Let's examine these aspects in greater detail:
Regioselectivity: Anti-Markovnikov Addition
As previously mentioned, hydroboration-oxidation displays anti-Markovnikov regioselectivity. This is a significant advantage over other hydration methods, such as acid-catalyzed hydration, which follow Markovnikov's rule. This characteristic allows for the synthesis of alcohols that would be difficult or impossible to obtain using other methods. The anti-Markovnikov addition is a direct consequence of the transition state during the hydroboration step, where steric hindrance plays a crucial role.
Stereoselectivity: Syn Addition
The reaction is also highly stereoselective, proceeding via syn addition. This means the hydrogen and hydroxyl group are added to the same face of the alkene. This syn stereoselectivity is a result of the concerted nature of the hydroboration step. The boron and hydrogen atom add simultaneously to the same side of the double bond, preventing the formation of diastereomers. This contrasts sharply with reactions like halohydrin formation, which proceed via anti addition.
Choosing the Right Borane Reagent: Optimizing the Reaction
The choice of borane reagent significantly impacts the reaction's efficiency and selectivity. Different borane derivatives offer varying degrees of steric hindrance and reactivity:
-
BH₃ (Borane): Highly reactive but can be challenging to handle due to its tendency to form dimers. Often used in THF solution.
-
9-BBN (9-Borabicyclo[3.3.1]nonane): A more bulky and selective reagent, particularly useful for sterically hindered alkenes. Offers improved regioselectivity compared to borane.
-
Disiamylborane: Another sterically hindered borane reagent, providing high selectivity.
The choice depends on the specific alkene substrate and desired selectivity. For simple alkenes, borane might suffice, while for more complex or sterically hindered alkenes, 9-BBN or disiamylborane are preferred for better control of regio- and stereochemistry.
Applications of Hydroboration-Oxidation: A Versatile Reaction
The hydroboration-oxidation reaction finds wide application in organic synthesis due to its unique properties:
-
Synthesis of Alcohols: It is a primary method for synthesizing alcohols, particularly those with anti-Markovnikov regiochemistry. This is crucial for preparing alcohols that are difficult to synthesize through other routes.
-
Preparation of Chiral Alcohols: Its high stereoselectivity makes it valuable in the asymmetric synthesis of chiral alcohols, providing a route to enantiomerically enriched products.
-
Functionalization of Alkenes: The resulting alcohol can be further functionalized to access a wide array of other functional groups.
-
Total Synthesis: The reaction often appears as a key step in complex organic syntheses of natural products and pharmaceuticals. The predictable regio- and stereochemistry makes it a reliable tool for complex molecule construction.
Limitations and Considerations
While hydroboration-oxidation is a powerful tool, it has some limitations:
-
Sensitivity to Oxygen and Moisture: Boranes are highly reactive towards oxygen and moisture, necessitating anhydrous conditions for optimal results. Careful handling and purification of reagents are essential.
-
Reactivity with Functional Groups: Boranes can react with other functional groups present in the molecule, leading to side reactions. Careful consideration of functional group compatibility is necessary.
-
Steric Hindrance: Highly sterically hindered alkenes might react slowly or inefficiently. The choice of borane reagent can mitigate this limitation to some extent.
Conclusion: A Powerful Tool in Organic Synthesis
The hydroboration-oxidation reaction is a valuable tool in organic synthesis, offering a reliable and efficient method for converting alkenes into alcohols with high regio- and stereoselectivity. Its anti-Markovnikov regioselectivity and syn stereochemistry make it a distinct and indispensable reaction in the arsenal of organic chemists. Understanding its mechanism, limitations, and the factors affecting its selectivity is crucial for successfully applying this powerful transformation in various synthetic endeavors. By carefully selecting the appropriate borane reagent and reaction conditions, chemists can leverage this reaction's unique properties to achieve desired synthetic outcomes, contributing to advancements in various fields of chemistry, including pharmaceuticals, materials science, and natural product synthesis. Continued research continues to expand the applications and refine the understanding of this pivotal reaction in modern organic chemistry.
Latest Posts
Latest Posts
-
Why Is The Light Microscope Also Called The Compound Microscope
Apr 06, 2025
-
What Is 15 4 As A Mixed Number
Apr 06, 2025
-
Sublimation Is Physical Or Chemical Change
Apr 06, 2025
-
Write 2 1 2 As An Improper Fraction
Apr 06, 2025
-
For Which Of The Mixtures Will Ag2so4 S Precipitate
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about Identify The Characteristics Of The Hydroboration-oxidation Of An Alkene . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
