How Many Pi Bonds Are In A Triple Bond
listenit
Apr 05, 2025 · 5 min read
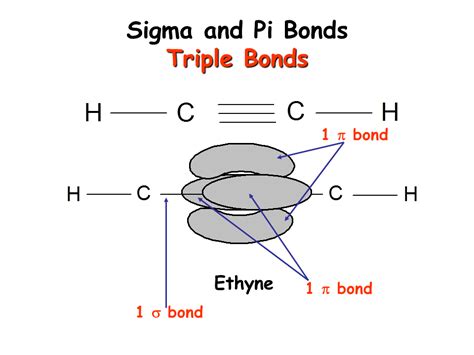
Table of Contents
How Many Pi Bonds Are in a Triple Bond? Understanding Chemical Bonding
The question of how many pi bonds are present in a triple bond is fundamental to understanding the nature of chemical bonding and the structure of molecules. This article delves deep into this concept, explaining the intricacies of sigma and pi bonds, their formation, and their significance in determining molecular properties. We'll explore various examples and discuss the implications of triple bonds in different chemical contexts.
Understanding Sigma (σ) and Pi (π) Bonds
Before we tackle the central question, let's establish a clear understanding of sigma and pi bonds. These are two types of covalent bonds, formed by the overlap of atomic orbitals. The key difference lies in the orientation of the overlapping orbitals.
Sigma Bonds (σ Bonds)
A sigma bond is formed by the head-on overlap of atomic orbitals. This means the electron density is concentrated along the internuclear axis—the imaginary line connecting the two bonded atoms. Sigma bonds are the strongest type of covalent bond and are always present in a single, double, or triple bond. Think of it as the foundational bond.
Pi Bonds (π Bonds)
A pi bond, on the other hand, is formed by the sideways overlap of p orbitals. The electron density is concentrated above and below the internuclear axis. Pi bonds are weaker than sigma bonds because the overlap of the p orbitals is less effective than the head-on overlap in sigma bonds. Importantly, pi bonds can only form after a sigma bond has already been established.
The Structure of a Triple Bond
Now, let's get to the heart of the matter: a triple bond. A triple bond is a type of covalent bond where three pairs of electrons are shared between two atoms. This bond is significantly stronger than a single or double bond due to the increased electron density and stronger attractive forces between the atoms.
Crucially, a triple bond always consists of one sigma (σ) bond and two pi (π) bonds. The sigma bond forms first through the head-on overlap of atomic orbitals (usually s and p orbitals), providing the fundamental framework for the bond. Then, two pi bonds are formed through the sideways overlap of two pairs of p orbitals, adding to the overall bond strength and impacting the molecule's geometry.
Visualizing the Triple Bond
Imagine two carbon atoms forming a triple bond. One carbon atom's s orbital and one of its p orbitals overlap head-on with the corresponding orbitals of the other carbon atom to form a sigma bond. The remaining two p orbitals on each carbon atom then overlap sideways to form two pi bonds, completing the triple bond. This arrangement is represented visually by showing the sigma bond as a single line and the two pi bonds as two parallel lines alongside it.
Examples of Triple Bonds in Chemistry
Triple bonds are common in several molecules, significantly impacting their properties and reactivity. Let's explore some key examples:
1. Alkynes
Alkynes are hydrocarbons containing at least one carbon-carbon triple bond. The simplest alkyne is ethyne (acetylene), with the formula C₂H₂. The triple bond between the two carbon atoms accounts for the alkyne's unique properties, including its high reactivity and linear geometry. Each carbon atom in ethyne is sp hybridized, meaning one s and one p orbital are involved in sigma bond formation, leaving two p orbitals to form two pi bonds.
2. Nitriles
Nitriles are organic compounds containing a cyano group (-CN), characterized by a triple bond between a carbon and a nitrogen atom. The nitrogen atom contributes one electron to the sigma bond and two electrons to the two pi bonds. This functional group imparts significant reactivity and unique spectroscopic properties to the molecule.
3. Carbon Monoxide (CO)
Carbon monoxide is a simple but crucial molecule featuring a triple bond between a carbon atom and an oxygen atom. This triple bond is incredibly strong and contributes to carbon monoxide's toxicity—its strong binding to the iron in hemoglobin prevents oxygen transport. The carbon and oxygen atoms are both sp hybridized.
4. Cyanogen (C₂N₂)
Cyanogen is another example featuring carbon-nitrogen triple bonds. The molecule consists of two carbon atoms linked to each other via a single bond, each of which is also connected to a nitrogen atom via a triple bond. This symmetrical structure provides a good illustration of the versatility of triple bond formation.
The Impact of Triple Bonds on Molecular Geometry and Properties
The presence of a triple bond significantly affects a molecule's geometry and properties:
-
Linear Geometry: Molecules with triple bonds tend to have a linear geometry around the atoms involved in the triple bond. This is due to the sp hybridization of the atoms participating in the triple bond.
-
High Bond Energy: Triple bonds are characterized by high bond energies due to the strong overlap of orbitals. This translates to greater stability and higher temperatures needed to break the bond.
-
Reactivity: Triple bonds exhibit distinct reactivity patterns compared to single and double bonds. The pi electrons are more exposed and readily participate in reactions like addition reactions.
Addressing Common Misconceptions
It’s crucial to clarify some common misconceptions surrounding triple bonds:
-
Triple bonds are not three sigma bonds: Only one sigma bond is always present; the additional two are pi bonds.
-
Triple bonds are not just stronger single bonds: The fundamental difference in the type of orbital overlap leads to the distinct chemical and physical properties associated with triple bonds.
-
The strength of a triple bond is not simply three times the strength of a single bond: While the presence of three bonds leads to increased strength, the specific energy and stability vary significantly depending on the atoms involved.
Conclusion: The Significance of Pi Bonds in Triple Bonds
In summary, a triple bond comprises one sigma (σ) bond and two pi (π) bonds. The pi bonds, formed by the sideways overlap of p orbitals, contribute significantly to the bond's strength, reactivity, and influence on molecular geometry. Understanding the fundamental distinctions between sigma and pi bonds and their roles in triple bond formation is essential for comprehending the behavior and properties of numerous organic and inorganic molecules. From alkynes to nitriles and carbon monoxide, the presence of a triple bond imparts unique characteristics that are crucial to diverse chemical applications and biological processes. The strong, linear, and reactive nature of the triple bond continues to be a central topic of study in chemical research and a key factor in the development of new materials and technologies.
Latest Posts
Latest Posts
-
What Is 5 To The Power Of 1
Apr 05, 2025
-
What Is 25 As A Fraction
Apr 05, 2025
-
What Is 12 Out Of 18
Apr 05, 2025
-
How To Calculate Molarity In Titration
Apr 05, 2025
-
How Do You Write 5 As A Decimal
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about How Many Pi Bonds Are In A Triple Bond . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
