How To Calculate Molarity In Titration
listenit
Apr 05, 2025 · 5 min read
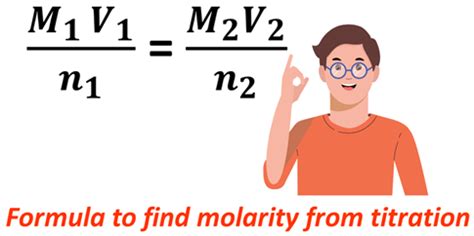
Table of Contents
How to Calculate Molarity in Titration: A Comprehensive Guide
Titration is a fundamental analytical technique in chemistry used to determine the concentration of an unknown solution, called the analyte, by reacting it with a solution of known concentration, called the titrant. The point at which the reaction is complete is called the equivalence point, and its precise determination is crucial for accurate molarity calculations. This comprehensive guide will walk you through the process of calculating molarity in titration, covering various aspects and providing clear examples.
Understanding the Fundamentals: Molarity and Titration
Before diving into the calculations, let's refresh our understanding of key concepts:
Molarity (M)
Molarity is a measure of concentration, defined as the number of moles of solute per liter of solution. The formula is:
Molarity (M) = Moles of solute / Liters of solution
Understanding molarity is essential because titration directly utilizes this concept to determine the unknown concentration.
Titration Process
Titration involves the gradual addition of a titrant from a burette to a known volume of analyte in a flask. An indicator is often used to visually signal the equivalence point, usually through a distinct color change. The key is to accurately measure the volume of titrant required to completely react with the analyte.
Types of Titration
Several types of titrations exist, each requiring slightly different approaches to calculations. Common types include:
- Acid-Base Titration: This involves reacting an acid with a base, using indicators sensitive to pH changes.
- Redox Titration: This involves reacting an oxidizing agent with a reducing agent.
- Precipitation Titration: This involves reacting two solutions to form a precipitate.
- Complexometric Titration: This involves the formation of a complex ion.
While the specific calculations might differ slightly, the underlying principles remain consistent. This guide will primarily focus on acid-base titrations, as they are the most commonly encountered type.
Calculating Molarity: A Step-by-Step Approach
The calculation of molarity in titration involves using the stoichiometry of the balanced chemical equation and the volumes and concentrations of the titrant and analyte. Let's break down the process step-by-step:
Step 1: Write a Balanced Chemical Equation
The first crucial step is to write a balanced chemical equation representing the reaction between the titrant and analyte. This equation provides the stoichiometric ratio between the reactants, essential for accurate calculations.
For example, in a titration of a strong acid (HCl) with a strong base (NaOH):
HCl(aq) + NaOH(aq) → NaCl(aq) + H₂O(l)
This equation shows a 1:1 mole ratio between HCl and NaOH. This means one mole of HCl reacts completely with one mole of NaOH.
Step 2: Determine the Moles of Titrant Used
Using the volume and molarity of the titrant, calculate the number of moles used in the titration. Remember to convert the volume from milliliters (mL) to liters (L) using the conversion factor 1 L = 1000 mL.
Moles of titrant = Molarity of titrant (M) × Volume of titrant (L)
Step 3: Determine the Moles of Analyte
Use the stoichiometric ratio from the balanced chemical equation to determine the moles of analyte that reacted with the titrant. This is where the 1:1, 2:1, or other mole ratios from the balanced equation become crucial.
Moles of analyte = Moles of titrant × (Stoichiometric ratio of analyte to titrant)
Step 4: Calculate the Molarity of the Analyte
Finally, calculate the molarity of the analyte using the moles of analyte and the initial volume of the analyte solution. Remember to convert the volume from milliliters (mL) to liters (L).
Molarity of analyte (M) = Moles of analyte / Volume of analyte (L)
Illustrative Examples
Let's solidify our understanding with a few examples:
Example 1: 1:1 Stoichiometry
25.00 mL of 0.100 M NaOH is used to titrate 20.00 mL of an HCl solution. Calculate the molarity of the HCl solution.
-
Balanced Equation: HCl(aq) + NaOH(aq) → NaCl(aq) + H₂O(l) (1:1 ratio)
-
Moles of NaOH: 0.100 M × (25.00 mL / 1000 mL/L) = 0.00250 moles NaOH
-
Moles of HCl: 0.00250 moles NaOH × (1 mole HCl / 1 mole NaOH) = 0.00250 moles HCl
-
Molarity of HCl: 0.00250 moles HCl / (20.00 mL / 1000 mL/L) = 0.125 M HCl
Example 2: Non-1:1 Stoichiometry
20.00 mL of 0.200 M KMnO₄ is used to titrate 15.00 mL of an Fe²⁺ solution according to the following balanced equation:
MnO₄⁻(aq) + 5Fe²⁺(aq) + 8H⁺(aq) → Mn²⁺(aq) + 5Fe³⁺(aq) + 4H₂O(l)
Calculate the molarity of the Fe²⁺ solution.
-
Balanced Equation: (Note the 1:5 ratio between MnO₄⁻ and Fe²⁺)
-
Moles of KMnO₄: 0.200 M × (20.00 mL / 1000 mL/L) = 0.00400 moles KMnO₄
-
Moles of Fe²⁺: 0.00400 moles KMnO₄ × (5 moles Fe²⁺ / 1 mole KMnO₄) = 0.0200 moles Fe²⁺
-
Molarity of Fe²⁺: 0.0200 moles Fe²⁺ / (15.00 mL / 1000 mL/L) = 1.33 M Fe²⁺
Dealing with Complications and Sources of Error
While the calculations appear straightforward, several factors can influence accuracy:
- Indicator Choice: The indicator used must accurately signal the equivalence point. Incorrect indicator selection can lead to significant errors.
- End-Point vs. Equivalence Point: The observed end-point might slightly differ from the true equivalence point. This difference is called indicator error.
- Incomplete Reactions: Reactions may not go to completion, especially in weak acid-weak base titrations, leading to inaccurate results.
- Improper Calibration of Equipment: Inaccurate measurements from the burette or pipette directly affect the calculated molarity.
- Parallax Error: Incorrect reading of the meniscus level in the burette can introduce errors.
Advanced Titration Calculations: Back Titration
Back titration is a useful technique when direct titration is difficult or impossible. It involves adding an excess of titrant to the analyte, allowing the reaction to proceed completely, and then titrating the remaining excess titrant with a second titrant. This technique is particularly helpful in cases involving slow reactions or insoluble analytes. The calculations for back titration involve determining the moles of excess titrant, subtracting this from the initial moles of titrant, and then proceeding with the molarity calculation as usual.
Conclusion
Calculating molarity in titration is a fundamental skill in analytical chemistry. Understanding the stoichiometry of the reaction, using accurate measurements, and properly applying the step-by-step procedure are crucial for obtaining reliable results. While various factors can affect the accuracy, careful attention to detail can minimize errors and ensure accurate determination of the analyte's molarity. Remember to always double-check your calculations and consider potential sources of error to refine your experimental technique and ensure the validity of your results. Consistent practice and attention to detail will build your proficiency in this critical analytical chemistry skill.
Latest Posts
Latest Posts
-
Federalists Supported Ratification Of The Us Constitution Because They
Apr 06, 2025
-
4 Protons 5 Neutrons 4 Electrons
Apr 06, 2025
-
What Is The Average Atomic Weight Of Silver
Apr 06, 2025
-
Find The Equivalent Resistance Between Points A And B
Apr 06, 2025
-
What Is 3 In Decimal Form
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about How To Calculate Molarity In Titration . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
