How Many Lone Pairs Does Co2 Have
listenit
Apr 02, 2025 · 5 min read
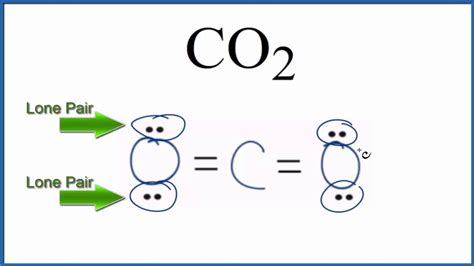
Table of Contents
How Many Lone Pairs Does CO₂ Have? Understanding Molecular Geometry and Electron Configuration
Carbon dioxide (CO₂) is a simple yet crucial molecule found throughout our environment and essential for various biological and industrial processes. Understanding its electronic structure, particularly the number of lone pairs, is key to comprehending its properties and reactivity. This article will delve deep into the electronic configuration of CO₂, explaining how to determine the number of lone pairs present and exploring the implications of its molecular geometry.
Determining the Number of Valence Electrons
Before we can determine the number of lone pairs, we need to understand the total number of valence electrons in the CO₂ molecule. Valence electrons are the outermost electrons in an atom, which participate in chemical bonding.
- Carbon (C): Carbon is in group 14 of the periodic table, meaning it has 4 valence electrons.
- Oxygen (O): Oxygen is in group 16, possessing 6 valence electrons each. Since there are two oxygen atoms, we have a total of 12 valence electrons from oxygen.
Therefore, the total number of valence electrons in CO₂ is 4 (from carbon) + 12 (from two oxygens) = 16 valence electrons.
Lewis Structure and Electron Pair Distribution
The Lewis structure is a visual representation of the molecule's bonding and lone pairs. To draw the Lewis structure for CO₂:
- Central Atom: Carbon, being less electronegative than oxygen, is placed in the center.
- Single Bonds: We initially connect each oxygen atom to the central carbon atom with a single bond. Each single bond uses two electrons. This accounts for 4 of the 16 valence electrons.
- Octet Rule: Oxygen atoms need to fulfill the octet rule (8 electrons in their valence shell). To achieve this, we add three lone pairs (6 electrons) to each oxygen atom. This accounts for 12 electrons (6 per oxygen atom).
At this stage, we have used all 16 valence electrons, but the central carbon atom only has 4 electrons, failing to satisfy the octet rule. To resolve this:
- Double Bonds: We convert one lone pair from each oxygen atom into a double bond with the carbon atom. This forms two double bonds (C=O), with each double bond using four electrons.
The final Lewis structure shows carbon double-bonded to each oxygen atom: O=C=O.
Lone Pairs in CO₂: The Answer
After forming the double bonds, we observe that each oxygen atom has two lone pairs (four electrons not involved in bonding). The carbon atom has zero lone pairs; all its four valence electrons are used in bonding.
Therefore, the answer is: CO₂ has a total of four lone pairs; two on each oxygen atom.
Molecular Geometry and its Implications
The molecular geometry of CO₂ is linear. This arises from the presence of two double bonds and the absence of lone pairs on the central carbon atom. The linear geometry influences various properties of CO₂:
- Polarity: Despite the polar nature of the C=O bonds (oxygen is more electronegative than carbon), the linear geometry results in a symmetrical distribution of charge. The dipole moments of the two C=O bonds cancel each other out, making CO₂ a nonpolar molecule.
- Boiling Point: The weak intermolecular forces (London dispersion forces) in nonpolar CO₂ result in a low boiling point (-78.5 °C).
- Solubility: CO₂'s nonpolar nature makes it relatively insoluble in polar solvents like water.
Comparing CO₂ with Other Molecules: Illustrating Lone Pair Concepts
Let's compare CO₂ with other molecules to further clarify the concept of lone pairs and their influence on molecular geometry:
1. Water (H₂O): Water has a central oxygen atom bonded to two hydrogen atoms. Oxygen has six valence electrons, two are used in bonding with hydrogen, leaving four electrons as two lone pairs on the oxygen atom. The two lone pairs influence the molecular geometry, resulting in a bent shape. This bent shape and the presence of lone pairs make water a polar molecule.
2. Ammonia (NH₃): Ammonia has a central nitrogen atom bonded to three hydrogen atoms. Nitrogen has five valence electrons; three are used in bonding, leaving two electrons as one lone pair on the nitrogen atom. This lone pair influences the molecular geometry, resulting in a trigonal pyramidal shape. The lone pair and the three N-H bonds make ammonia a polar molecule.
3. Methane (CH₄): Methane has a central carbon atom bonded to four hydrogen atoms. Carbon has four valence electrons, all of which are used in bonding. Therefore, methane has zero lone pairs. Its molecular geometry is tetrahedral, and it's a nonpolar molecule.
Advanced Concepts: Hybridization and Molecular Orbital Theory
While the Lewis structure provides a simplified representation, a more accurate description of bonding in CO₂ involves concepts like hybridization and molecular orbital theory.
-
Hybridization: Carbon in CO₂ undergoes sp hybridization. One s orbital and one p orbital combine to form two sp hybrid orbitals, which are involved in sigma bonding with the oxygen atoms. The remaining two p orbitals on carbon participate in pi bonding with oxygen.
-
Molecular Orbital Theory: This theory provides a more sophisticated picture of bonding by describing molecular orbitals formed from the combination of atomic orbitals. In CO₂, sigma and pi bonding molecular orbitals are formed, leading to a stable molecule.
Conclusion: Understanding the Significance of Lone Pairs
The seemingly simple question of how many lone pairs CO₂ possesses actually opens the door to understanding crucial concepts in chemistry. By determining the number of valence electrons, constructing the Lewis structure, and considering molecular geometry, we can accurately depict the molecule's electronic structure. This understanding allows us to predict and explain various properties of CO₂, its reactivity, and its crucial role in various natural and industrial processes. Understanding lone pairs isn't just about counting electrons; it's the key to unlocking the deeper secrets of molecular structure and behavior. The comparison with other molecules further solidifies the importance of lone pairs in determining molecular geometry and polarity, impacting the molecule's physical and chemical properties. Finally, the introduction of hybridization and molecular orbital theory offers a more complete and accurate understanding of the bonding within CO₂.
Latest Posts
Latest Posts
-
How Is A Mole Similar To A Dozen
Apr 03, 2025
-
Which Is The Aqueous And Organic Layer
Apr 03, 2025
-
Is Soh Cah Toa Only For Right Triangles
Apr 03, 2025
-
How To Calculate The Heat Of Neutralization
Apr 03, 2025
-
What Are The Rows In The Periodic Table Called
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about How Many Lone Pairs Does Co2 Have . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
