How To Calculate The Heat Of Neutralization
listenit
Apr 03, 2025 · 6 min read
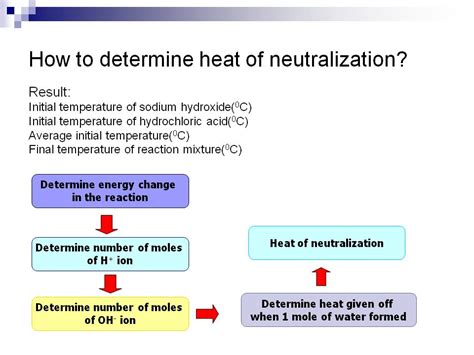
Table of Contents
How to Calculate the Heat of Neutralization: A Comprehensive Guide
The heat of neutralization, also known as the enthalpy of neutralization, is the heat change that occurs when an acid and a base react to form one mole of water. Understanding how to calculate this value is crucial in various fields, including chemistry, chemical engineering, and thermodynamics. This comprehensive guide will walk you through the process, explaining the concepts, methods, and considerations involved.
Understanding the Fundamentals
Before diving into the calculations, let's solidify our understanding of the underlying principles. Neutralization reactions are exothermic, meaning they release heat. This heat release is due to the formation of strong bonds between hydrogen ions (H⁺) from the acid and hydroxide ions (OH⁻) from the base to form water (H₂O). The heat of neutralization is typically expressed in kilojoules per mole (kJ/mol).
The heat released depends on several factors, including:
- The strength of the acid and base: Strong acids and bases completely dissociate in water, leading to a more significant heat release compared to weak acids and bases. Weak acids and bases only partially dissociate, resulting in less heat being released. The heat of neutralization for a strong acid-strong base reaction is generally around -57 kJ/mol. Variations occur due to differences in ionic interactions.
- The concentration of the reactants: Higher concentrations lead to a more substantial heat change because more moles of reactants are involved.
- The temperature of the reactants: The initial temperature affects the final temperature change and, consequently, the calculated heat of neutralization.
- The solvent: While water is the most common solvent, using different solvents can alter the heat of neutralization.
Methods for Calculating Heat of Neutralization
There are several methods to determine the heat of neutralization experimentally and subsequently calculate the value. The most common method involves calorimetry.
1. Using Calorimetry: The Coffee Cup Calorimeter
A coffee cup calorimeter is a simple yet effective apparatus for measuring heat changes in solution. It's based on the principle of heat transfer between the reacting system and its surroundings. The reaction takes place inside the calorimeter, and the temperature change is monitored. The heat absorbed or released by the reaction is calculated based on the temperature change, the specific heat capacity of the solution, and the mass of the solution.
Step-by-Step Procedure:
-
Prepare the solutions: Measure precise volumes of the acid and base solutions using volumetric glassware. Ensure the solutions are at room temperature before mixing. The concentrations should be known and relatively equal. Consider using a strong acid and a strong base for more consistent results.
-
Measure initial temperature: Carefully measure the temperature of both solutions using a thermometer. Take an average temperature if there is a slight difference.
-
Mix the solutions: Add the acid solution to the calorimeter. Then, carefully add the base solution to the acid. Stir gently but thoroughly to ensure complete mixing.
-
Monitor the temperature change: Continuously monitor the temperature of the mixture using the thermometer. Record the highest or lowest temperature reached, depending on whether the reaction is exothermic or endothermic (neutralization reactions are typically exothermic).
-
Calculate the heat change: Use the following formula:
q = mcΔT
Where:
- q = heat absorbed or released (in Joules)
- m = mass of the solution (in grams) – This is approximately the sum of the masses of the acid and base solutions, assuming the densities are close to that of water (1 g/mL).
- c = specific heat capacity of the solution (usually assumed to be close to the specific heat capacity of water, 4.18 J/g°C)
- ΔT = change in temperature (final temperature - initial temperature)
-
Calculate the moles of water formed: Determine the number of moles of water produced from the stoichiometry of the balanced neutralization reaction. For example, in the reaction HCl + NaOH → NaCl + H₂O, one mole of HCl reacts with one mole of NaOH to produce one mole of water.
-
Calculate the heat of neutralization: Divide the heat change (q) by the number of moles of water formed. The result is the heat of neutralization (ΔH) in Joules per mole. Convert to kilojoules per mole by dividing by 1000.
Example:
Let's say we mixed 50 mL of 1.0 M HCl and 50 mL of 1.0 M NaOH in a calorimeter. The initial temperature was 25°C, and the final temperature was 31°C.
- m ≈ 100 g (assuming density of 1 g/mL)
- c ≈ 4.18 J/g°C
- ΔT = 31°C - 25°C = 6°C
q = (100 g)(4.18 J/g°C)(6°C) = 2508 J
Moles of water formed = 0.05 L × 1.0 mol/L = 0.05 mol
ΔH = 2508 J / 0.05 mol = 50160 J/mol = 50.16 kJ/mol
This value is an approximation due to heat loss to the surroundings.
2. Using Bomb Calorimetry
Bomb calorimetry provides a more accurate measurement of heat changes, especially for reactions that are not carried out in solution. However, it is more complex and requires specialized equipment. Bomb calorimetry is more precise in accounting for heat loss.
3. Using Hess's Law
Hess's Law states that the enthalpy change for a reaction is independent of the pathway taken. This principle can be utilized to calculate the heat of neutralization indirectly if the heats of formation of the reactants and products are known. It allows for the calculation of ΔH for the neutralization reaction using standard enthalpy of formation data. This is done by summing the standard enthalpies of formation of the products and subtracting the sum of the standard enthalpies of formation of the reactants. Databases of standard enthalpy of formation values are widely available.
Sources of Error and Mitigation Strategies
Several factors can introduce errors into the heat of neutralization calculation:
- Heat loss to the surroundings: Heat is inevitably lost to the air and the calorimeter itself. This leads to an underestimation of the actual heat released. Minimizing heat loss is crucial and can be done by using insulated calorimeters, covering the calorimeter, and ensuring rapid mixing to reduce reaction time.
- Incomplete mixing: Failure to completely mix the acid and base leads to inaccurate temperature measurements. Thorough and gentle stirring minimizes this.
- Heat capacity of the calorimeter: The calorimeter itself absorbs some heat. This can be accounted for by calibrating the calorimeter using a known reaction with a known heat change.
- Incomplete reaction: If the acid or base is weak, the reaction may not go to completion. This will affect the heat of neutralization calculated. Using strong acids and bases ensures a more complete reaction.
- Evaporation: The loss of water due to evaporation can affect the concentration and mass of the solution, thereby influencing the results.
Applications of Heat of Neutralization
The heat of neutralization has practical applications in various fields:
- Chemical Engineering: Design of reactors, heat exchangers, and other chemical processes.
- Environmental Science: Understanding neutralization reactions in wastewater treatment.
- Thermodynamics: Studying the thermodynamics of acid-base reactions.
Conclusion
Calculating the heat of neutralization requires careful experimental design and execution. The coffee cup calorimeter method provides a relatively simple and accessible approach, while bomb calorimetry offers greater accuracy. Understanding the sources of error and implementing mitigation strategies is vital for obtaining reliable results. The value of the heat of neutralization provides valuable information for understanding acid-base reactions and their thermodynamic properties, making it a critical concept in various scientific and engineering disciplines. Remember to always wear appropriate safety equipment when conducting experiments involving chemicals. Always consult your teacher or supervisor before undertaking any laboratory work.
Latest Posts
Latest Posts
-
Lowest Common Multiple Of 2 3 And 7
Apr 03, 2025
-
What Is The Decimal Of 8 9
Apr 03, 2025
-
How Are Heat And Temperature Related
Apr 03, 2025
-
37 1 2 As A Fraction
Apr 03, 2025
-
How Many Ounes In A Pound
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about How To Calculate The Heat Of Neutralization . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
