How Are Elements Arranged In The Modern Periodic Table
listenit
Apr 03, 2025 · 6 min read
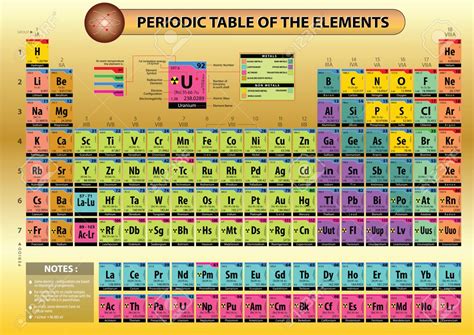
Table of Contents
How Are Elements Arranged in the Modern Periodic Table?
The modern periodic table, a cornerstone of chemistry, isn't just a random arrangement of elements. Its highly organized structure reflects the fundamental properties of atoms and their relationships. Understanding this arrangement is key to grasping the behavior of matter and predicting chemical reactions. This article delves into the intricacies of the modern periodic table, explaining the principles behind its organization and showcasing its predictive power.
From Mendeleev's Vision to the Modern Table
Before the modern periodic table, scientists struggled to organize the growing list of known elements. Dmitri Mendeleev, a Russian chemist, is credited with creating the first periodic table in 1869. He arranged elements in order of increasing atomic weight, noticing recurring patterns in their properties. This arrangement, although imperfect by today's standards, was a revolutionary step, allowing Mendeleev to predict the existence and properties of undiscovered elements.
Mendeleev's genius lay in leaving gaps in his table for undiscovered elements, predicting their properties based on the trends observed in surrounding elements. This predictive power solidified the importance of his work. Later discoveries confirmed his predictions, cementing the periodic table's validity and its ability to organize chemical knowledge.
However, Mendeleev's table wasn't perfect. Some elements were placed out of order based solely on atomic weight to maintain the pattern of properties. This discrepancy hinted at a more fundamental organizing principle.
The Atomic Number: The Key to Understanding
The advent of modern physics and the discovery of the atomic nucleus clarified the underlying principle governing the periodic table: atomic number. The atomic number, represented by Z, is the number of protons in an atom's nucleus. This is the crucial factor determining an element's properties. Unlike atomic weight, which can vary due to isotopes (atoms of the same element with different numbers of neutrons), the atomic number is unique to each element.
The modern periodic table arranges elements in increasing order of their atomic number. This seemingly simple change solved the inconsistencies present in Mendeleev's table, providing a more accurate and robust framework for understanding chemical behavior.
Periods and Groups: Unveiling the Patterns
The periodic table is structured into periods (rows) and groups (columns). The arrangement within these periods and groups reveals recurring patterns in the electronic configuration of atoms, directly impacting their chemical properties.
Periods: Reflecting Electron Shells
Each period corresponds to a principal energy level or electron shell. As you move across a period, electrons are added sequentially to the outermost electron shell. This increase in electrons across a period affects the element's properties. For example, elements on the left side of a period tend to be metals, while those on the right are nonmetals, with a gradual transition in properties between them. The number of elements in a period varies, reflecting the complexities of electron shell filling.
Groups: Sharing Similar Chemical Properties
Groups, or families, are the vertical columns of the periodic table. Elements within the same group share similar chemical properties because they have the same number of valence electrons. Valence electrons are the electrons in the outermost shell, directly involved in chemical bonding. This similarity in valence electron configuration dictates how elements react with other elements, forming compounds with predictable characteristics.
For example, the alkali metals (Group 1) all have one valence electron, making them highly reactive and readily losing that electron to form +1 ions. The halogens (Group 17) have seven valence electrons and readily gain one electron to form -1 ions. This consistent behavior within groups is a powerful tool for predicting chemical reactions.
Exploring the Blocks: s, p, d, and f
The periodic table can be further divided into blocks, based on the subshells into which the valence electrons are added. These blocks are labeled s, p, d, and f, reflecting the shapes of the orbitals involved.
s-block elements: Alkali and Alkaline Earth Metals
The s-block elements occupy the first two groups of the periodic table. They are characterized by valence electrons in the s subshell. The alkali metals (Group 1) and alkaline earth metals (Group 2) are found here, demonstrating predictable trends in reactivity and ionic charge.
p-block elements: Diverse Properties
The p-block elements occupy groups 13-18. Their valence electrons are in the p subshell. This block encompasses a wide range of elements with diverse properties, including both metals and nonmetals, as well as metalloids (elements with intermediate properties between metals and nonmetals).
d-block elements: Transition Metals
The d-block elements, located in the middle of the periodic table, are known as transition metals. They are characterized by partially filled d subshells, giving them variable oxidation states and often resulting in colorful compounds. The transition metals are known for their catalytic properties and diverse applications in various industries.
f-block elements: Lanthanides and Actinides
The f-block elements, also known as the inner transition metals, are usually placed separately at the bottom of the periodic table. They are divided into the lanthanides and actinides, characterized by partially filled f subshells. The f-block elements exhibit complex electronic configurations and subtle variations in their properties. Many actinides are radioactive.
Predicting Properties: The Power of the Periodic Table
The beauty of the periodic table lies in its predictive power. By understanding the trends and patterns within the table, chemists can predict various properties of elements and their compounds. These predictions encompass:
- Atomic Radius: Generally, atomic radius increases down a group and decreases across a period.
- Ionization Energy: The energy required to remove an electron from an atom increases across a period and decreases down a group.
- Electronegativity: The ability of an atom to attract electrons in a chemical bond increases across a period and decreases down a group.
- Metallic Character: Metallic character generally increases down a group and decreases across a period.
- Reactivity: The reactivity of metals tends to increase down a group and decrease across a period. The reactivity of nonmetals tends to increase across a period and decrease down a group.
Beyond the Basics: Modern Refinements
While the basic principles outlined above form the foundation of the periodic table, ongoing research continues to refine our understanding and extend its application. For example:
- Isotopes: The concept of isotopes and their varying atomic weights adds a layer of complexity, emphasizing that the atomic number is the truly defining characteristic of an element.
- Electron Configuration: A deeper understanding of electron configuration and its relationship to chemical properties enhances the predictive power of the periodic table.
- Computational Chemistry: Modern computational tools allow for detailed modeling of atomic and molecular properties, further enriching our understanding and predicting behavior beyond simple trends.
Conclusion: A Dynamic and Evolving System
The modern periodic table isn't a static entity; it's a constantly evolving system that continues to shape our understanding of chemistry. Its elegant structure, based on the fundamental principle of atomic number and reflected in the periodic trends, provides a framework for understanding the properties of elements and predicting their behavior. This ability to predict and interpret chemical phenomena has made the periodic table indispensable in various fields, from materials science to medicine, highlighting its enduring importance in the scientific community and beyond. The periodic table remains a testament to the power of observation, prediction, and the relentless pursuit of scientific knowledge.
Latest Posts
Latest Posts
-
How Many Valence Electrons In Al
Apr 04, 2025
-
Silver Has A Density Of 10 5 G Cm3
Apr 04, 2025
-
Common Factor Of 32 And 36
Apr 04, 2025
-
How To Find Mass Of A Planet
Apr 04, 2025
-
What Is Molar Volume Of Gas At Stp
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about How Are Elements Arranged In The Modern Periodic Table . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
