Explain The Relationship Between Monomers And Polymers
listenit
Apr 02, 2025 · 7 min read
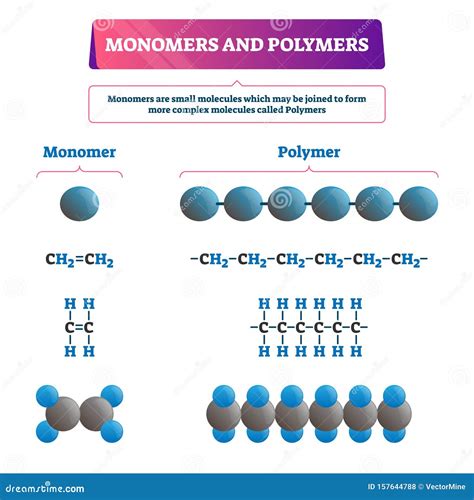
Table of Contents
The Amazing World of Monomers and Polymers: A Deep Dive into their Relationship
The world around us is built from incredibly tiny building blocks. From the DNA in our cells to the plastic bottles in our recycling bins, the fundamental principle of polymerization—the linking of smaller units into larger chains—is at play. Understanding the relationship between monomers and polymers is key to understanding the properties of countless materials, both natural and synthetic. This article will explore this fundamental relationship in detail, covering everything from the basic definitions to the diverse applications of polymeric materials.
What are Monomers?
Monomers are the individual building blocks of polymers. The word itself is derived from the Greek words "mono" (one) and "meros" (part). These are relatively small molecules, often containing reactive functional groups that allow them to bond with other monomers. Think of them as individual Lego bricks—incapable of forming a complex structure on their own, but crucial for building something larger and more intricate.
The chemical nature of a monomer dictates the properties of the resulting polymer. Different monomers have different chemical structures, leading to a vast array of polymer types with vastly different characteristics. Some common examples of monomers include:
- Ethylene (CH₂=CH₂): The simplest alkene and the monomer of polyethylene, a widely used plastic.
- Styrene (C₈H₈): Used to produce polystyrene, commonly found in packaging and disposable cups.
- Vinyl chloride (C₂H₃Cl): The monomer of polyvinyl chloride (PVC), a versatile plastic used in pipes, flooring, and many other applications.
- Glucose (C₆H₁₂O₆): A simple sugar and the monomer of cellulose and starch, essential components of plant cell walls and energy storage in plants, respectively.
- Amino acids: The building blocks of proteins, crucial for biological functions. Each amino acid has a unique side chain that contributes to the overall protein structure and function.
Functional Groups: The Key to Polymerization
Monomers often possess specific functional groups—reactive parts of the molecule—that enable them to form chemical bonds with other monomers. These functional groups can include:
- Alkenes (C=C double bonds): These are common in many synthetic polymers, undergoing addition polymerization.
- Carboxylic acids (-COOH): Involved in the formation of polyesters and polyamides through condensation polymerization.
- Alcohols (-OH): Also involved in condensation polymerization, often reacting with carboxylic acids.
- Amines (-NH₂): Participate in the formation of polyamides (like nylon) and other polymers.
What are Polymers?
Polymers are macromolecules composed of many repeating monomer units. The word "polymer" originates from the Greek words "poly" (many) and "meros" (part). These long chains can consist of thousands or even millions of monomer units linked together. This long chain structure gives polymers unique properties, often very different from their individual monomer units. Think of the intricate Lego castle created from those simple bricks—the final product possesses qualities far beyond those of a single brick.
Types of Polymerization: Building the Chains
Two main types of polymerization processes lead to the formation of polymers:
-
Addition Polymerization: This type involves the direct addition of monomers to a growing polymer chain without the loss of any atoms. The monomers typically contain a double bond (like alkenes) that opens up to form single bonds with other monomers. Examples include polyethylene, polypropylene, and polyvinyl chloride (PVC).
-
Condensation Polymerization: In this process, monomers react to form a polymer chain, releasing a small molecule (often water) as a byproduct. This involves the reaction of functional groups, such as carboxylic acids and alcohols, to form ester linkages. Examples include polyesters (like PET) and polyamides (like nylon).
Properties of Polymers: A Diverse Range
The properties of polymers are remarkably diverse, influenced by several factors including:
- The type of monomer: Different monomers result in different polymer properties, from flexible and elastic to rigid and strong.
- The length of the polymer chain: Longer chains generally lead to stronger and more durable materials.
- The degree of branching: Branching can affect the polymer's density, flexibility, and melting point.
- Cross-linking: Cross-linking between polymer chains increases strength and rigidity.
- The presence of additives: Additives can be incorporated to modify the polymer's properties, such as improving its flexibility, color, or resistance to heat or UV light.
The Relationship Between Monomers and Polymers: A Closer Look
The relationship between monomers and polymers is fundamentally one of building blocks and structure. The properties of a polymer are directly related to the type, arrangement, and interactions of its monomer units. A small change in the monomer structure can have a dramatic effect on the final polymer's characteristics.
For instance, consider the difference between polyethylene (PE) and polypropylene (PP). Both are addition polymers, but PE is made from ethylene monomers, while PP uses propylene monomers. This seemingly small difference leads to significant variations in properties: PE is more flexible and less dense than PP, which is stronger and more rigid.
The arrangement of monomers within the polymer chain also matters. Linear polymers, with monomers arranged in a straight chain, tend to be more flexible, while branched or cross-linked polymers are typically stronger and more resistant to heat.
Examples of Monomer-Polymer Relationships:
-
Natural Polymers: DNA, a crucial genetic material, is a polymer made from nucleotide monomers. The sequence of these nucleotides determines the genetic code. Proteins, essential for countless biological processes, are polymers built from amino acid monomers. The specific sequence and folding of these amino acids determine the protein's function. Cellulose, a major component of plant cell walls, is a polymer of glucose monomers.
-
Synthetic Polymers: Polyethylene terephthalate (PET), a common plastic used in bottles and clothing fibers, is a polyester formed by the condensation polymerization of ethylene glycol and terephthalic acid monomers. Polyvinyl chloride (PVC), used in pipes and flooring, is an addition polymer of vinyl chloride monomers. Nylon, a strong and durable synthetic fiber, is a polyamide formed by the condensation polymerization of diamines and diacids.
Applications of Polymers: A Vast and Growing Field
The applications of polymers are virtually limitless, spanning numerous industries:
-
Packaging: Polymers like polyethylene (PE), polypropylene (PP), and polyethylene terephthalate (PET) are widely used in food packaging, due to their low cost, ease of processing, and ability to provide a barrier to moisture and oxygen.
-
Construction: Polymers are used in building materials such as pipes, insulation, and roofing membranes, providing durability, lightweight properties, and resistance to corrosion.
-
Textiles: Polymers such as nylon, polyester, and acrylic are used to create synthetic fibers for clothing, carpets, and other textiles. These fibers offer various properties such as strength, elasticity, and wrinkle resistance.
-
Automotive: Polymers are incorporated into various automotive components, such as bumpers, dashboards, and interior parts, providing lightweight, durable, and aesthetically pleasing features.
-
Medicine: Biocompatible polymers are employed in medical devices such as implants, drug delivery systems, and artificial organs.
-
Electronics: Polymers are used in electronics applications, such as insulation in wires, printed circuit boards, and flexible displays.
The Future of Monomers and Polymers: Innovations and Sustainability
Research into monomers and polymers continues to advance rapidly, driving innovation in materials science and engineering. This includes the development of:
-
Bio-based polymers: These polymers are derived from renewable resources such as plants, reducing reliance on fossil fuels.
-
Biodegradable polymers: These polymers break down naturally in the environment, minimizing pollution.
-
High-performance polymers: These polymers exhibit superior properties, such as high strength, heat resistance, and chemical resistance, opening new possibilities in various applications.
-
Smart polymers: These polymers respond to environmental stimuli such as temperature, pH, or light, enabling applications in areas like drug delivery and sensors.
The understanding and manipulation of the relationship between monomers and polymers is crucial for developing sustainable and advanced materials that address the needs of a growing global population. As research progresses, we can expect to see even more innovative applications of polymers, shaping the future of various industries and improving our lives in countless ways. From the microscopic world of cellular processes to the macroscopic scale of building construction, the power of polymerization remains a fundamental force shaping our world.
Latest Posts
Latest Posts
-
How Many Electrons Can Fit In The First Energy Level
Apr 03, 2025
-
The Basic Unit Of A Chemical Element
Apr 03, 2025
-
What Happens To The Temperature During A Phase Change
Apr 03, 2025
-
How To Find Relative Minimum And Maximum
Apr 03, 2025
-
2 3 Or 3 4 Bigger
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Explain The Relationship Between Monomers And Polymers . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
