During A Chemical Reaction Atoms Are
listenit
Apr 05, 2025 · 6 min read
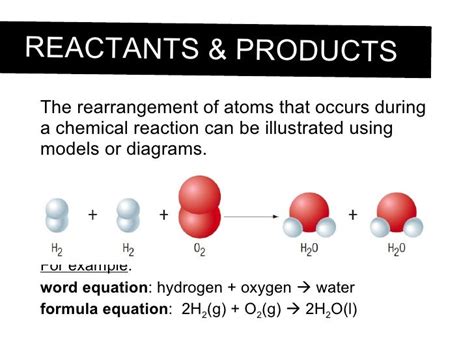
Table of Contents
During a Chemical Reaction, Atoms Are… Rearranged! A Deep Dive into Atomic Behavior
Chemical reactions are the fundamental building blocks of the universe, driving everything from the rusting of iron to the processes of life itself. But what exactly happens at the atomic level during these reactions? The short answer is: atoms are rearranged. This seemingly simple statement, however, belies a complex and fascinating world of electron interactions, bond breaking, and bond formation. This article will delve into the intricacies of atomic behavior during chemical reactions, exploring the underlying principles and providing illustrative examples.
The Indestructible Atom: The Law of Conservation of Mass
A cornerstone of chemistry is the Law of Conservation of Mass, which states that matter cannot be created or destroyed in a chemical reaction. This means that the total mass of the reactants (the starting materials) equals the total mass of the products (the substances formed). While atoms can be rearranged and combined in new ways, they themselves remain intact. No atoms disappear, and no new atoms spontaneously appear. This fundamental principle governs all chemical reactions.
Implications of Conservation of Mass
The Law of Conservation of Mass has profound implications for our understanding of chemical reactions. It allows us to balance chemical equations, predicting the quantities of reactants needed and products formed. This predictive power is essential in various fields, from industrial chemistry to environmental science. It allows chemists to efficiently design experiments and processes, minimizing waste and maximizing yield.
The Dance of Electrons: Chemical Bonds and Reactions
Chemical reactions are essentially a reshuffling of atoms, driven by the interactions of their outermost electrons – the valence electrons. These electrons are involved in the formation of chemical bonds, which are the forces that hold atoms together in molecules and compounds. There are several types of chemical bonds, including:
1. Ionic Bonds: The Transfer of Electrons
Ionic bonds form through the transfer of electrons from one atom to another. This transfer creates ions: positively charged cations (atoms that have lost electrons) and negatively charged anions (atoms that have gained electrons). The electrostatic attraction between these oppositely charged ions forms the ionic bond. A classic example is the formation of sodium chloride (table salt) from sodium (Na) and chlorine (Cl). Sodium readily loses one electron to achieve a stable electron configuration, becoming a Na⁺ ion. Chlorine readily gains one electron, becoming a Cl⁻ ion. The electrostatic attraction between Na⁺ and Cl⁻ forms the ionic bond in NaCl.
2. Covalent Bonds: The Sharing of Electrons
Covalent bonds are formed by the sharing of electrons between atoms. This sharing allows each atom to achieve a stable electron configuration, fulfilling the octet rule (eight electrons in the outermost shell for most atoms). Many organic molecules, like water (H₂O) and methane (CH₄), are held together by covalent bonds. In water, oxygen shares electrons with two hydrogen atoms, while in methane, carbon shares electrons with four hydrogen atoms.
3. Metallic Bonds: A Sea of Electrons
Metallic bonds are found in metals and involve the delocalization of valence electrons. These electrons are not associated with any particular atom but rather move freely throughout the metal lattice, creating a "sea" of electrons. This delocalization accounts for the characteristic properties of metals, such as high electrical and thermal conductivity, malleability, and ductility.
Breaking and Making Bonds: The Mechanism of Reaction
During a chemical reaction, existing chemical bonds are broken, and new bonds are formed. This process involves changes in the energy of the system. Energy is required to break bonds (bond dissociation energy), while energy is released when new bonds are formed (bond formation energy).
Activation Energy: The Energy Barrier
The activation energy is the minimum energy required to initiate a chemical reaction. It represents the energy barrier that must be overcome for the reactants to reach a transition state, where bonds are broken and new bonds begin to form. Once this energy barrier is surpassed, the reaction proceeds, often releasing energy in the process (exothermic reaction) or requiring further energy input (endothermic reaction).
Reaction Mechanisms: A Step-by-Step Approach
Complex reactions often proceed through a series of intermediate steps, known as a reaction mechanism. These mechanisms describe the precise sequence of bond breaking and bond formation events that lead to the formation of the products. Understanding reaction mechanisms is crucial for predicting reaction rates and designing catalysts to speed up reactions.
Examples of Atomic Rearrangement in Chemical Reactions
Let's consider a few specific examples to illustrate how atoms are rearranged during chemical reactions:
1. Combustion of Methane:
The combustion of methane (CH₄) with oxygen (O₂) produces carbon dioxide (CO₂) and water (H₂O):
CH₄ + 2O₂ → CO₂ + 2H₂O
In this reaction, the covalent bonds in methane and oxygen are broken, and new covalent bonds are formed to create carbon dioxide and water. The carbon atom initially bonded to four hydrogen atoms now forms double bonds with two oxygen atoms. The hydrogen atoms, previously bonded to carbon, now form single bonds with oxygen atoms to create water molecules.
2. Formation of Ammonia:
The Haber-Bosch process synthesizes ammonia (NH₃) from nitrogen (N₂) and hydrogen (H₂):
N₂ + 3H₂ → 2NH₃
This reaction involves the breaking of the strong triple bond in nitrogen and the single bonds in hydrogen. New covalent bonds are then formed between nitrogen and hydrogen atoms to produce ammonia molecules. This reaction is crucial for fertilizer production and highlights the significance of atomic rearrangement in industrial processes.
3. Neutralization Reaction:
The reaction between an acid and a base is a neutralization reaction. For example, the reaction between hydrochloric acid (HCl) and sodium hydroxide (NaOH) produces sodium chloride (NaCl) and water (H₂O):
HCl + NaOH → NaCl + H₂O
Here, ionic bonds are broken in both the acid and the base. The hydrogen ion (H⁺) from the acid combines with the hydroxide ion (OH⁻) from the base to form water. The sodium ion (Na⁺) and the chloride ion (Cl⁻) then form an ionic bond to create sodium chloride.
Beyond Chemical Reactions: Nuclear Reactions
It's important to distinguish between chemical reactions and nuclear reactions. While chemical reactions involve the rearrangement of atoms and their electrons, nuclear reactions involve changes in the nucleus of the atom. Nuclear reactions can result in the formation of different isotopes or even entirely different elements. These reactions involve vastly larger energy changes compared to chemical reactions and are not governed by the same principles.
Conclusion: The Dynamic World of Atoms
In conclusion, during a chemical reaction, atoms are not created or destroyed but rather rearranged. This rearrangement involves the breaking and forming of chemical bonds, driven by the interactions of valence electrons. Understanding this fundamental principle is crucial for comprehending the vast array of chemical phenomena that shape our world. From the synthesis of life-sustaining molecules to the development of new materials, the rearrangement of atoms is the engine of change. The careful study of these processes provides insights into the intricate dance of atoms and their profound impact on the world around us. The laws governing these rearrangements are fundamental to chemistry and provide a powerful framework for understanding the universe. Further exploration into reaction mechanisms, kinetics, and thermodynamics will reveal even more about the dynamic world of atoms in action.
Latest Posts
Latest Posts
-
How Many Protons Are In A Sulfur Atom
Apr 05, 2025
-
What Is The Square Root Of 2 25
Apr 05, 2025
-
What Is The Least Common Multiple Of 9 And 3
Apr 05, 2025
-
The Horizontal Columns On The Periodic Table Are Called
Apr 05, 2025
-
What Is The Least Common Multiple Of 14 And 7
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about During A Chemical Reaction Atoms Are . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
