How Many Protons Are In A Sulfur Atom
listenit
Apr 05, 2025 · 6 min read
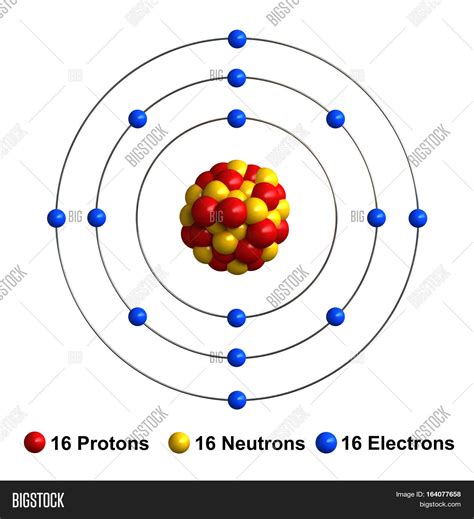
Table of Contents
How Many Protons Are in a Sulfur Atom? Understanding Atomic Structure and Sulfur's Properties
Determining the number of protons in a sulfur atom is fundamental to understanding its chemical behavior and its place within the periodic table. This seemingly simple question opens the door to a deeper exploration of atomic structure, isotopes, and the fascinating world of chemistry. Let's delve into this topic, uncovering not only the answer but also the broader implications it holds.
Understanding Atomic Structure: The Foundation of Chemistry
Before we pinpoint the number of protons in sulfur, let's establish a basic understanding of atomic structure. An atom, the fundamental building block of matter, is composed of three primary subatomic particles:
- Protons: Positively charged particles found within the atom's nucleus. The number of protons defines the element itself. It's the atomic number.
- Neutrons: Neutrally charged particles also residing in the nucleus. They contribute to the atom's mass but not its charge. The number of neutrons can vary within an element, leading to isotopes.
- Electrons: Negatively charged particles orbiting the nucleus in electron shells or energy levels. The number of electrons usually equals the number of protons in a neutral atom.
The nucleus, the dense central core of the atom, contains both protons and neutrons. The electrons, being significantly lighter, occupy the space surrounding the nucleus. This arrangement dictates an atom's chemical properties and interactions with other atoms.
The Atomic Number: Identifying Elements
The key to identifying an element lies in its atomic number, which represents the number of protons in the nucleus of its atoms. This number is unique to each element and is universally recognized as a fundamental property. You'll find the atomic number listed on the periodic table, usually above the element's symbol.
The periodic table is a cornerstone of chemistry, organizing elements based on their atomic structure and properties. Its arrangement reveals recurring trends and relationships between elements, enabling predictions about their chemical behavior.
Sulfur: A Closer Look at its Atomic Structure
Sulfur (S), a nonmetal with atomic number 16, is an abundant element found in various forms on Earth. Its unique electronic configuration and chemical reactivity make it crucial in various biological and industrial processes.
So, to answer the central question: a sulfur atom contains 16 protons. This is its defining characteristic, differentiating it from all other elements.
Isotopes of Sulfur: Variations in Neutron Count
While the number of protons remains constant for a given element, the number of neutrons can vary, creating isotopes. Isotopes of an element share the same atomic number (proton count) but have different mass numbers (total number of protons and neutrons).
Sulfur has several naturally occurring isotopes, including:
- Sulfur-32 (³²S): The most abundant isotope, containing 16 protons and 16 neutrons.
- Sulfur-33 (³³S): A less abundant isotope with 16 protons and 17 neutrons.
- Sulfur-34 (³⁴S): Another naturally occurring isotope, containing 16 protons and 18 neutrons.
- Sulfur-36 (³⁶S): A rare, stable isotope with 16 protons and 20 neutrons.
These isotopes exhibit similar chemical behavior but differ slightly in mass, leading to minor variations in their physical properties. The relative abundance of each isotope affects the average atomic mass of sulfur, which is listed on the periodic table.
Sulfur's Role in Biology and Industry: A Versatile Element
Sulfur's chemical properties, a direct consequence of its 16 protons and electron configuration, make it essential for various processes.
Biological Significance:
- Amino Acids: Sulfur is a crucial component of certain amino acids, such as cysteine and methionine, which are the building blocks of proteins. These proteins are essential for numerous biological functions.
- Enzymes: Many enzymes, the biological catalysts that speed up chemical reactions, contain sulfur atoms in their active sites. Their functioning relies on the presence of sulfur.
- Vitamins: Certain vitamins, like biotin (vitamin B7), contain sulfur and play a vital role in metabolism.
Industrial Applications:
- Sulfuric Acid: Sulfuric acid (H₂SO₄), a highly corrosive and reactive acid, is a cornerstone of many industrial processes. It is used in fertilizer production, oil refining, and metal processing, to name a few.
- Rubber Vulcanization: Sulfur is crucial in the vulcanization of rubber, a process that improves its strength, durability, and elasticity. This process revolutionized the rubber industry.
- Match Heads: Historically, sulfur played a role in the production of match heads due to its flammability. Modern matches often still contain sulfur compounds.
- Pharmaceuticals: Sulfur compounds find use in various pharmaceuticals, contributing to drug efficacy and delivery.
Beyond Protons: The Significance of Electron Configuration
While the number of protons defines the element, the electron configuration determines its chemical reactivity. Sulfur's 16 electrons are arranged in specific energy levels or shells around the nucleus, dictating its tendency to gain or share electrons to achieve a stable electron configuration.
This tendency leads to sulfur's ability to form various chemical bonds, contributing to its versatility in forming numerous compounds. Understanding its electron configuration is crucial to comprehending its chemical behavior and its interactions with other elements.
Connecting the Dots: From Protons to Chemical Properties
The seemingly simple fact that sulfur has 16 protons is foundational to its entire chemical persona. This number dictates its atomic number, placing it precisely in the periodic table. It affects its isotopic variations and, most importantly, governs its electron configuration. The electron configuration, in turn, determines its reactivity and the types of bonds it can form.
Therefore, the 16 protons aren't just a numerical value; they're the cornerstone of sulfur's unique chemical identity. It shapes its behavior in biological systems, its industrial applications, and its overall contribution to the fascinating world of chemistry.
Exploring Further: Advanced Concepts Related to Sulfur
This exploration has covered the basics. However, for a deeper dive into sulfur's properties and behavior, consider investigating these advanced concepts:
- Sulfur's allotropes: Sulfur exists in various forms, known as allotropes, with different structural arrangements and physical properties.
- Sulfur's oxidation states: Sulfur can exist in multiple oxidation states, indicating its ability to gain or lose electrons in chemical reactions.
- Sulfur in environmental chemistry: Sulfur plays a role in environmental processes, including the formation of acid rain and the sulfur cycle.
- Advanced spectroscopic techniques for studying sulfur: Techniques like X-ray photoelectron spectroscopy (XPS) and nuclear magnetic resonance (NMR) spectroscopy allow detailed study of sulfur's electronic structure and chemical environment.
Conclusion: The Significance of a Simple Number
The answer to "How many protons are in a sulfur atom?" – 16 – is deceptively simple. However, this number unveils a wealth of information about the element's structure, behavior, and significance. From its role in biological molecules to its industrial applications, sulfur's properties are a direct consequence of its atomic number. Understanding this fundamental aspect of atomic structure helps us appreciate the intricate relationships between the subatomic world and the macroscopic properties of matter. It's a testament to the elegance and power of chemistry's underlying principles.
Latest Posts
Latest Posts
-
2x 1 2x 3 X 2
Apr 06, 2025
-
Function Of A Fruit In A Plant
Apr 06, 2025
-
What Number Is 0 5 Of 8
Apr 06, 2025
-
What Is The Difference Between Balanced Forces And Action Reaction Forces
Apr 06, 2025
-
The Atomic Number Is Equal To The
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about How Many Protons Are In A Sulfur Atom . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
