Ch4 O2 Co2 H2o Balance Equation
listenit
Apr 04, 2025 · 5 min read
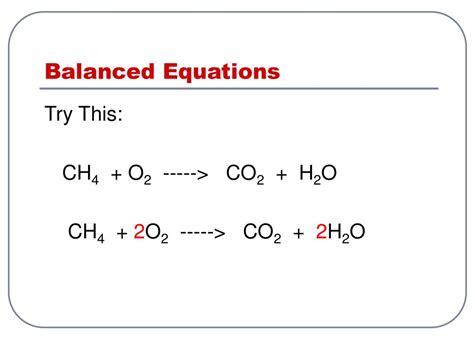
Table of Contents
Balancing the Equation: A Deep Dive into CH₄ + O₂ → CO₂ + H₂O
The combustion of methane (CH₄), a primary component of natural gas, is a fundamental chemical process with significant implications for energy production, atmospheric chemistry, and environmental science. Understanding the balanced chemical equation for this reaction – CH₄ + O₂ → CO₂ + H₂O – is crucial for comprehending its stoichiometry, energy release, and environmental impact. This article will delve into the intricacies of balancing this equation, exploring the underlying principles and practical applications.
Understanding the Reactants and Products
Before we balance the equation, let's understand the components involved:
Methane (CH₄)
Methane is a simple hydrocarbon, consisting of one carbon atom bonded to four hydrogen atoms. It's a colorless, odorless gas that is highly flammable and a potent greenhouse gas. Its role as a fuel source makes understanding its combustion crucial.
Oxygen (O₂)
Oxygen is a diatomic gas, essential for combustion. It acts as the oxidizing agent, reacting with methane to release energy. The availability of oxygen directly influences the completeness of the combustion reaction.
Carbon Dioxide (CO₂)
Carbon dioxide is the primary product of complete methane combustion. It's a greenhouse gas, contributing to global warming. The amount of CO₂ produced is directly proportional to the amount of methane burned.
Water (H₂O)
Water is another product of complete methane combustion. While not a greenhouse gas in the same way as CO₂, the production of water vapor can have implications for atmospheric humidity and climate.
Balancing the Equation: A Step-by-Step Guide
Balancing a chemical equation ensures that the number of atoms of each element is equal on both the reactant and product sides. This adheres to the law of conservation of mass, stating that matter cannot be created or destroyed in a chemical reaction. Here's how to balance the methane combustion equation:
1. Start with the most complex molecule: In this case, methane (CH₄) is the most complex molecule.
2. Balance Carbon (C): There's one carbon atom on the reactant side (in CH₄) and one carbon atom on the product side (in CO₂). Carbon is already balanced.
3. Balance Hydrogen (H): There are four hydrogen atoms on the reactant side (in CH₄) and two hydrogen atoms on the product side (in H₂O). To balance hydrogen, we need to place a coefficient of 2 in front of H₂O:
CH₄ + O₂ → CO₂ + 2H₂O
4. Balance Oxygen (O): Now, let's balance oxygen. There are two oxygen atoms on the reactant side (in O₂) and four oxygen atoms on the product side (two from CO₂ and two from 2H₂O). To balance oxygen, we need to place a coefficient of 2 in front of O₂:
CH₄ + 2O₂ → CO₂ + 2H₂O
The Balanced Equation: The balanced equation for the complete combustion of methane is:
CH₄ + 2O₂ → CO₂ + 2H₂O
Implications of the Balanced Equation
The balanced equation provides crucial information:
-
Stoichiometry: The equation shows the molar ratios of reactants and products. For every one mole of methane reacted, two moles of oxygen are required, producing one mole of carbon dioxide and two moles of water.
-
Energy Release: The combustion of methane is an exothermic reaction, meaning it releases energy in the form of heat. The balanced equation helps calculate the amount of heat released using thermochemical data. This is crucial for designing and optimizing combustion engines and power plants.
-
Environmental Impact: The equation highlights the production of greenhouse gases (CO₂) during methane combustion. Understanding the stoichiometry allows for estimations of CO₂ emissions from various sources, aiding in climate change mitigation strategies.
-
Incomplete Combustion: If the supply of oxygen is limited, incomplete combustion occurs, producing carbon monoxide (CO) and soot (carbon particles) in addition to CO₂ and H₂O. The balanced equation for complete combustion doesn't represent these scenarios. Incomplete combustion is far more dangerous due to the toxicity of CO.
Variations and Considerations
Several factors can affect the actual combustion process and deviate from the idealized balanced equation:
-
Temperature and Pressure: The reaction rate and equilibrium are influenced by temperature and pressure conditions.
-
Presence of other substances: Impurities in the methane or the presence of catalysts can affect the reaction pathway and products.
-
Reaction kinetics: The speed at which the reaction proceeds depends on factors like activation energy and surface area.
Practical Applications
The understanding of the balanced equation for methane combustion is fundamental in several fields:
-
Energy Production: Power plants use methane combustion to generate electricity. The balanced equation is essential for efficient fuel utilization and emissions control.
-
Industrial Processes: Methane combustion is used in various industrial processes requiring high temperatures, such as manufacturing and chemical synthesis.
-
Environmental Monitoring: The balanced equation helps in analyzing emissions from various sources and developing strategies to reduce greenhouse gas emissions.
-
Climate Modeling: Accurate modeling of climate change requires precise understanding of methane combustion and its contribution to atmospheric CO₂ levels.
Advanced Topics: Beyond the Basics
While the basic balanced equation provides a solid foundation, a deeper understanding requires considering more advanced concepts:
-
Thermochemistry: This branch of chemistry deals with the heat changes during chemical reactions. Using enthalpy of formation data, one can calculate the heat released during methane combustion.
-
Chemical Kinetics: This area focuses on the reaction rates and mechanisms. Understanding the kinetics helps optimize combustion processes for efficiency and minimize pollutant formation.
-
Equilibrium Considerations: While the reaction generally proceeds to completion under typical conditions, understanding equilibrium constants can be important in specific scenarios.
-
Computational Chemistry: Computational methods are increasingly used to simulate and analyze combustion reactions at the molecular level, providing insights into reaction mechanisms and pathways.
Conclusion
The seemingly simple balanced equation for the combustion of methane – CH₄ + 2O₂ → CO₂ + 2H₂O – is a cornerstone of understanding a crucial chemical process with wide-ranging applications and environmental implications. From energy production to climate modeling, mastering this equation and its underlying principles is essential for addressing numerous challenges in science, technology, and society. The deeper you delve into this reaction, the more you appreciate the interconnectedness of chemistry, energy, and the environment. Further exploration into thermochemistry, kinetics, and computational methods will reveal even more nuanced understanding of this fundamental reaction.
Latest Posts
Latest Posts
-
How Do You Find Ml Quantum Number
Apr 04, 2025
-
How Many Unpaired Electrons Does Manganese Have
Apr 04, 2025
-
Water Is Made Up Of Which Two Elements
Apr 04, 2025
-
What Is 9 Percent Of 50
Apr 04, 2025
-
Distance From Earth To Pluto In Light Years
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Ch4 O2 Co2 H2o Balance Equation . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
